+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-8011 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Foot region of the yeast spliceosomal U4/U6.U5 tri-snRNP | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Transcription / pre-mRNA splicing / snRNP / GTPase / U5 snRNA / Prp8 / spliceosome | |||||||||
| Function / homology |  Function and homology information Function and homology informationgeneration of catalytic spliceosome for first transesterification step / splicing factor binding / U4/U6 snRNP / spliceosome conformational change to release U4 (or U4atac) and U1 (or U11) / 7-methylguanosine cap hypermethylation / snRNP binding / pICln-Sm protein complex / small nuclear ribonucleoprotein complex / SMN-Sm protein complex / spliceosomal tri-snRNP complex ...generation of catalytic spliceosome for first transesterification step / splicing factor binding / U4/U6 snRNP / spliceosome conformational change to release U4 (or U4atac) and U1 (or U11) / 7-methylguanosine cap hypermethylation / snRNP binding / pICln-Sm protein complex / small nuclear ribonucleoprotein complex / SMN-Sm protein complex / spliceosomal tri-snRNP complex / poly(U) RNA binding / commitment complex / U2-type prespliceosome assembly / U2-type catalytic step 2 spliceosome / U4 snRNP / U2 snRNP / U1 snRNP / U2-type prespliceosome / precatalytic spliceosome / generation of catalytic spliceosome for second transesterification step / spliceosomal complex assembly / mRNA 5'-splice site recognition / mRNA 3'-splice site recognition / spliceosomal tri-snRNP complex assembly / U5 snRNA binding / U5 snRNP / U2 snRNA binding / U6 snRNA binding / spliceosomal snRNP assembly / pre-mRNA intronic binding / U1 snRNA binding / U4/U6 x U5 tri-snRNP complex / catalytic step 2 spliceosome / spliceosomal complex / mRNA splicing, via spliceosome / metallopeptidase activity / GTPase activity / mRNA binding / GTP binding / RNA binding / nucleus / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |  | |||||||||
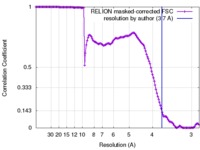
| Method | single particle reconstruction / Resolution: 3.7 Å | |||||||||
 Authors Authors | Nguyen THD / Galej WP | |||||||||
| Funding support |  United Kingdom, 1 items United Kingdom, 1 items
| |||||||||
 Citation Citation |  Journal: Nature / Year: 2015 Journal: Nature / Year: 2015Title: The architecture of the spliceosomal U4/U6.U5 tri-snRNP. Authors: Thi Hoang Duong Nguyen / Wojciech P Galej / Xiao-chen Bai / Christos G Savva / Andrew J Newman / Sjors H W Scheres / Kiyoshi Nagai /  Abstract: U4/U6.U5 tri-snRNP is a 1.5-megadalton pre-assembled spliceosomal complex comprising U5 small nuclear RNA (snRNA), extensively base-paired U4/U6 snRNAs and more than 30 proteins, including the key ...U4/U6.U5 tri-snRNP is a 1.5-megadalton pre-assembled spliceosomal complex comprising U5 small nuclear RNA (snRNA), extensively base-paired U4/U6 snRNAs and more than 30 proteins, including the key components Prp8, Brr2 and Snu114. The tri-snRNP combines with a precursor messenger RNA substrate bound to U1 and U2 small nuclear ribonucleoprotein particles (snRNPs), and transforms into a catalytically active spliceosome after extensive compositional and conformational changes triggered by unwinding of the U4 and U6 (U4/U6) snRNAs. Here we use cryo-electron microscopy single-particle reconstruction of Saccharomyces cerevisiae tri-snRNP at 5.9 Å resolution to reveal the essentially complete organization of its RNA and protein components. The single-stranded region of U4 snRNA between its 3' stem-loop and the U4/U6 snRNA stem I is loaded into the Brr2 helicase active site ready for unwinding. Snu114 and the amino-terminal domain of Prp8 position U5 snRNA to insert its loop I, which aligns the exons for splicing, into the Prp8 active site cavity. The structure provides crucial insights into the activation process and the active site of the spliceosome. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_8011.map.gz emd_8011.map.gz | 196.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-8011-v30.xml emd-8011-v30.xml emd-8011.xml emd-8011.xml | 27.6 KB 27.6 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_8011_fsc.xml emd_8011_fsc.xml | 13.3 KB | Display |  FSC data file FSC data file |
| Images |  emd_8011.png emd_8011.png | 150.6 KB | ||
| Filedesc metadata |  emd-8011.cif.gz emd-8011.cif.gz | 8.3 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-8011 http://ftp.pdbj.org/pub/emdb/structures/EMD-8011 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-8011 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-8011 | HTTPS FTP |
-Validation report
| Summary document |  emd_8011_validation.pdf.gz emd_8011_validation.pdf.gz | 266.8 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_8011_full_validation.pdf.gz emd_8011_full_validation.pdf.gz | 265.9 KB | Display | |
| Data in XML |  emd_8011_validation.xml.gz emd_8011_validation.xml.gz | 12.9 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-8011 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-8011 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-8011 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-8011 | HTTPS FTP |
-Related structure data
| Related structure data |  5gamMC  8006C  8007C  8008C  8009C  8010C  8012C  8013C  8014C  5ganC  5gaoC  5gapC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_8011.map.gz / Format: CCP4 / Size: 209.3 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_8011.map.gz / Format: CCP4 / Size: 209.3 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 1.43 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
+Entire : Foot region of the yeast spliceosomal U4/U6.U5 tri-snRNP
+Supramolecule #1: Foot region of the yeast spliceosomal U4/U6.U5 tri-snRNP
+Macromolecule #1: U5 snRNA
+Macromolecule #4: U6 snRNA, 5' end
+Macromolecule #2: Pre-mRNA-splicing factor 8
+Macromolecule #3: Pre-mRNA-splicing factor SNU114
+Macromolecule #5: Small nuclear ribonucleoprotein-associated protein B
+Macromolecule #6: Small nuclear ribonucleoprotein E
+Macromolecule #7: Small nuclear ribonucleoprotein F
+Macromolecule #8: Small nuclear ribonucleoprotein G
+Macromolecule #9: Unknown polypeptide
+Macromolecule #10: Small nuclear ribonucleoprotein Sm D3
+Macromolecule #11: Small nuclear ribonucleoprotein Sm D1
+Macromolecule #12: Small nuclear ribonucleoprotein Sm D2
+Macromolecule #13: GUANOSINE-5'-TRIPHOSPHATE
-Experimental details
-Structure determination
 Processing Processing | single particle reconstruction |
|---|---|
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.2 mg/mL | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.9 Component:
| |||||||||||||||
| Grid | Model: Quantifoil R1.2/1.3 / Material: COPPER / Mesh: 400 / Support film - Material: CARBON / Support film - topology: CONTINUOUS / Support film - Film thickness: 6 / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 70 sec. / Pretreatment - Atmosphere: OTHER Details: Grids are made of holey carbon, carbon-coated and glow discharged in N-amylamine. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Specialist optics | Energy filter - Name: GIF Quantum |
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: SUPER-RESOLUTION / Digitization - Frames/image: 1-20 / Number real images: 2477 / Average exposure time: 16.0 sec. / Average electron dose: 38.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated magnification: 35714 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.0 mm / Nominal defocus max: 3.5 µm / Nominal defocus min: 0.5 µm / Nominal magnification: 81000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Space: RECIPROCAL |
|---|---|
| Output model |  PDB-5gam: |
 Movie
Movie Controller
Controller