[English] 日本語
 Yorodumi
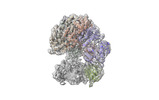
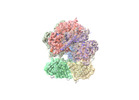
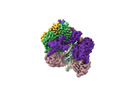
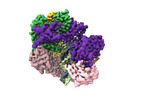
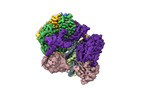
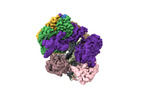
Yorodumi- EMDB-42406: Cryo-EM map of the human CTF18-RFC alone in the apo state (State 1) -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM map of the human CTF18-RFC alone in the apo state (State 1) | |||||||||
 Map data Map data | Primary sharpen EM map | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | DNA clamp loader complex /  REPLICATION REPLICATION | |||||||||
| Function / homology |  Function and homology information Function and homology informationresponse to organophosphorus / Ctf18 RFC-like complex / Rad17 RFC-like complex / Elg1 RFC-like complex / DNA replication factor C complex / positive regulation of DNA-directed DNA polymerase activity / Polymerase switching / DNA clamp loader activity / Polymerase switching on the C-strand of the telomere / single-stranded DNA helicase activity ...response to organophosphorus / Ctf18 RFC-like complex / Rad17 RFC-like complex / Elg1 RFC-like complex / DNA replication factor C complex / positive regulation of DNA-directed DNA polymerase activity / Polymerase switching / DNA clamp loader activity / Polymerase switching on the C-strand of the telomere / single-stranded DNA helicase activity / DNA duplex unwinding / DNA strand elongation involved in DNA replication / HDR through Single Strand Annealing (SSA) / Impaired BRCA2 binding to RAD51 / DNA synthesis involved in DNA repair / Presynaptic phase of homologous DNA pairing and strand exchange / PCNA-Dependent Long Patch Base Excision Repair / ATP-dependent activity, acting on DNA / Activation of ATR in response to replication stress / Translesion synthesis by REV1 / Translesion synthesis by POLK / Translesion synthesis by POLI / Gap-filling DNA repair synthesis and ligation in GG-NER / Recognition of DNA damage by PCNA-containing replication complex / Termination of translesion DNA synthesis / Translesion Synthesis by POLH / HDR through Homologous Recombination (HRR) / G2/M DNA damage checkpoint / Dual Incision in GG-NER / DNA-templated DNA replication / Dual incision in TC-NER / Gap-filling DNA repair synthesis and ligation in TC-NER / Processing of DNA double-strand break ends / Regulation of TP53 Activity through Phosphorylation /  DNA replication / DNA replication /  cell cycle / cell cycle /  DNA repair / DNA repair /  enzyme binding / enzyme binding /  ATP hydrolysis activity / ATP hydrolysis activity /  DNA binding / DNA binding /  nucleoplasm / nucleoplasm /  ATP binding / ATP binding /  membrane / membrane /  nucleus / nucleus /  cytosol cytosolSimilarity search - Function | |||||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method |  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 3.35 Å cryo EM / Resolution: 3.35 Å | |||||||||
 Authors Authors | Wang F / He Q / Li H | |||||||||
| Funding support |  United States, 2 items United States, 2 items
| |||||||||
 Citation Citation |  Journal: Proc Natl Acad Sci U S A / Year: 2024 Journal: Proc Natl Acad Sci U S A / Year: 2024Title: Cryo-EM reveals a nearly complete PCNA loading process and unique features of the human alternative clamp loader CTF18-RFC. Authors: Qing He / Feng Wang / Michael E O'Donnell / Huilin Li /  Abstract: The DNA sliding clamp PCNA is a multipurpose platform for DNA polymerases and many other proteins involved in DNA metabolism. The topologically closed PCNA ring needs to be cracked open and loaded ...The DNA sliding clamp PCNA is a multipurpose platform for DNA polymerases and many other proteins involved in DNA metabolism. The topologically closed PCNA ring needs to be cracked open and loaded onto DNA by a clamp loader, e.g., the well-studied pentameric ATPase complex RFC (RFC1-5). The CTF18-RFC complex is an alternative clamp loader found recently to bind the leading strand DNA polymerase ε and load PCNA onto leading strand DNA, but its structure and the loading mechanism have been unknown. By cryo-EM analysis of in vitro assembled human CTF18-RFC-DNA-PCNA complex, we have captured seven loading intermediates, revealing a detailed PCNA loading mechanism onto a 3'-ss/dsDNA junction by CTF18-RFC. Interestingly, the alternative loader has evolved a highly mobile CTF18 AAA+ module likely to lower the loading activity, perhaps to avoid competition with the RFC and to limit its role to leading strand clamp loading. To compensate for the lost stability due to the mobile AAA+ module, CTF18 has evolved a unique β-hairpin motif that reaches across RFC2 to interact with RFC5, thereby stabilizing the pentameric complex. Further, we found that CTF18 also contains a separation pin to locally melt DNA from the 3'-end of the primer; this ensures its ability to load PCNA to any 3'-ss/dsDNA junction, facilitated by the binding energy of the E-plug to the major groove. Our study reveals unique structural features of the human CTF18-RFC and contributes to a broader understanding of PCNA loading by the alternative clamp loaders. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_42406.map.gz emd_42406.map.gz | 117.9 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-42406-v30.xml emd-42406-v30.xml emd-42406.xml emd-42406.xml | 25.9 KB 25.9 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_42406_fsc.xml emd_42406_fsc.xml | 10.6 KB | Display |  FSC data file FSC data file |
| Images |  emd_42406.png emd_42406.png | 59.7 KB | ||
| Filedesc metadata |  emd-42406.cif.gz emd-42406.cif.gz | 7.4 KB | ||
| Others |  emd_42406_additional_1.map.gz emd_42406_additional_1.map.gz emd_42406_additional_2.map.gz emd_42406_additional_2.map.gz emd_42406_half_map_1.map.gz emd_42406_half_map_1.map.gz emd_42406_half_map_2.map.gz emd_42406_half_map_2.map.gz | 62.2 MB 111.1 MB 116.1 MB 116.2 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-42406 http://ftp.pdbj.org/pub/emdb/structures/EMD-42406 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-42406 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-42406 | HTTPS FTP |
-Related structure data
| Related structure data |  8unjMC  8umtC  8umuC  8umvC  8umwC  8umyC  8un0C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_42406.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_42406.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Primary sharpen EM map | ||||||||||||||||||||
| Voxel size | X=Y=Z: 0.828 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: Original unsharpen EM map
| File | emd_42406_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Original unsharpen EM map | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: Sharpen EM map by DeepEMhancer
| File | emd_42406_additional_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Sharpen EM map by DeepEMhancer | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: EM half map A
| File | emd_42406_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | EM half map A | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: EM half map B
| File | emd_42406_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | EM half map B | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : the human clamp loader CTF18_RFC
| Entire | Name: the human clamp loader CTF18_RFC |
|---|---|
| Components |
|
-Supramolecule #1: the human clamp loader CTF18_RFC
| Supramolecule | Name: the human clamp loader CTF18_RFC / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#5 |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 300 KDa |
-Macromolecule #1: Chromosome transmission fidelity protein 18 homolog
| Macromolecule | Name: Chromosome transmission fidelity protein 18 homolog / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 107.523984 KDa |
| Recombinant expression | Organism: Insect cell expression vector pTIE1 (others) |
| Sequence | String: MEDYEQELCG VEDDFHNQFA AELEVLAELE GASTPSPSGV PLFTAGRPPR TFEEALARGD AASSPAPAAS VGSSQGGARK RQVDADLQP AGSLPHAPRI KRPRLQVVKR LNFRSEEMEE PPPPDSSPTD ITPPPSPEDL AELWGHGVSE AAADVGLTRA S PAARNPVL ...String: MEDYEQELCG VEDDFHNQFA AELEVLAELE GASTPSPSGV PLFTAGRPPR TFEEALARGD AASSPAPAAS VGSSQGGARK RQVDADLQP AGSLPHAPRI KRPRLQVVKR LNFRSEEMEE PPPPDSSPTD ITPPPSPEDL AELWGHGVSE AAADVGLTRA S PAARNPVL RRPPILEDYV HVTSTEGVRA YLVLRADPMA PGVQGSLLHV PWRGGGQLDL LGVSLASLKK QVDGERRERL LQ EAQKLSD TLHSLRSGEE EAAQPLGAPE EEPTDGQDAS SHCLWVDEFA PRHYTELLSD DFTNRCLLKW LKLWDLVVFG HER PSRKPR PSVEPARVSK EATAPGKWKS HEQVLEEMLE AGLDPSQRPK QKVALLCGPP GLGKTTLAHV IARHAGYSVV EMNA SDDRS PEVFRTRIEA ATQMESVLGA GGKPNCLVID EIDGAPVAAI NVLLSILNRK GPQEVGPQGP AVPSGGGRRR RAEGG LLMR PIICICNDQF APSLRQLKQQ AFLLHFPPTL PSRLVQRLQE VSLRQGMRAD PGVLAALCEK TDNDIRACIN TLQFLY SRG QRELSVRDVQ ATRVGLKDQR RGLFSVWQEV FQLPRAQRRR VGQDPALPAD TLLLGDGDAG SLTSASQRFY RVLHAAA SA GEHEKVVQGL FDNFLRLRLR DSSLGAVCVA LDWLAFDDLL AGAAHHSQSF QLLRYPPFLP VAFHVLFASS HTPRITFP S SQQEAQNRMS QMRNLIQTLV SGIAPATRSR ATPQALLLDA LCLLLDILAP KLRPVSTQLY STREKQQLAS LVGTMLAYS LTYRQERTPD GQYIYRLEPN VEELCRFPEL PARKPLTYQT KQLIAREIEV EKMRRAEASA RVENSPQVDG SPPGLEGLLG GIGEKGVHR PAPRNHEQRL EHIMRRAARE EQPEKDFFGR VVVRSTAVPS AGDTAPEQDS VERRMGTAVG RSEVWFRFNE G VSNAVRRS LYIRDLL UniProtKB: Chromosome transmission fidelity protein 18 homolog |
-Macromolecule #2: Replication factor C subunit 2
| Macromolecule | Name: Replication factor C subunit 2 / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 39.203207 KDa |
| Recombinant expression | Organism: Insect cell expression vector pTIE1 (others) |
| Sequence | String: MEVEAVCGGA GEVEAQDSDP APAFSKAPGS AGHYELPWVE KYRPVKLNEI VGNEDTVSRL EVFAREGNVP NIIIAGPPGT GKTTSILCL ARALLGPALK DAMLELNASN DRGIDVVRNK IKMFAQQKVT LPKGRHKIII LDEADSMTDG AQQALRRTME I YSKTTRFA ...String: MEVEAVCGGA GEVEAQDSDP APAFSKAPGS AGHYELPWVE KYRPVKLNEI VGNEDTVSRL EVFAREGNVP NIIIAGPPGT GKTTSILCL ARALLGPALK DAMLELNASN DRGIDVVRNK IKMFAQQKVT LPKGRHKIII LDEADSMTDG AQQALRRTME I YSKTTRFA LACNASDKII EPIQSRCAVL RYTKLTDAQI LTRLMNVIEK ERVPYTDDGL EAIIFTAQGD MRQALNNLQS TF SGFGFIN SENVFKVCDE PHPLLVKEMI QHCVNANIDE AYKILAHLWH LGYSPEDIIG NIFRVCKTFQ MAEYLKLEFI KEI GYTHMK IAEGVNSLLQ MAGLLARLCQ KTMAPVAS UniProtKB:  Replication factor C subunit 2 Replication factor C subunit 2 |
-Macromolecule #3: Replication factor C subunit 5
| Macromolecule | Name: Replication factor C subunit 5 / type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 38.545512 KDa |
| Recombinant expression | Organism: Insect cell expression vector pTIE1 (others) |
| Sequence | String: METSALKQQE QPAATKIRNL PWVEKYRPQT LNDLISHQDI LSTIQKFINE DRLPHLLLYG PPGTGKTSTI LACAKQLYKD KEFGSMVLE LNASDDRGID IIRGPILSFA STRTIFKKGF KLVILDEADA MTQDAQNALR RVIEKFTENT RFCLICNYLS K IIPALQSR ...String: METSALKQQE QPAATKIRNL PWVEKYRPQT LNDLISHQDI LSTIQKFINE DRLPHLLLYG PPGTGKTSTI LACAKQLYKD KEFGSMVLE LNASDDRGID IIRGPILSFA STRTIFKKGF KLVILDEADA MTQDAQNALR RVIEKFTENT RFCLICNYLS K IIPALQSR CTRFRFGPLT PELMVPRLEH VVEEEKVDIS EDGMKALVTL SSGDMRRALN ILQSTNMAFG KVTEETVYTC TG HPLKSDI ANILDWMLNQ DFTTAYRNIT ELKTLKGLAL HDILTEIHLF VHRVDFPSSV RIHLLTKMAD IEYRLSVGTN EKI QLSSLI AAFQVTRDLI VAEA UniProtKB:  Replication factor C subunit 5 Replication factor C subunit 5 |
-Macromolecule #4: Replication factor C subunit 4
| Macromolecule | Name: Replication factor C subunit 4 / type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 39.735711 KDa |
| Recombinant expression | Organism: Insect cell expression vector pTIE1 (others) |
| Sequence | String: MQAFLKGTSI STKPPLTKDR GVAASAGSSG ENKKAKPVPW VEKYRPKCVD EVAFQEEVVA VLKKSLEGAD LPNLLFYGPP GTGKTSTIL AAARELFGPE LFRLRVLELN ASDERGIQVV REKVKNFAQL TVSGSRSDGK PCPPFKIVIL DEADSMTSAA Q AALRRTME ...String: MQAFLKGTSI STKPPLTKDR GVAASAGSSG ENKKAKPVPW VEKYRPKCVD EVAFQEEVVA VLKKSLEGAD LPNLLFYGPP GTGKTSTIL AAARELFGPE LFRLRVLELN ASDERGIQVV REKVKNFAQL TVSGSRSDGK PCPPFKIVIL DEADSMTSAA Q AALRRTME KESKTTRFCL ICNYVSRIIE PLTSRCSKFR FKPLSDKIQQ QRLLDIAKKE NVKISDEGIA YLVKVSEGDL RK AITFLQS ATRLTGGKEI TEKVITDIAG VIPAEKIDGV FAACQSGSFD KLEAVVKDLI DEGHAATQLV NQLHDVVVEN NLS DKQKSI ITEKLAEVDK CLADGADEHL QLISLCATVM QQLSQNC UniProtKB:  Replication factor C subunit 4 Replication factor C subunit 4 |
-Macromolecule #5: Replication factor C subunit 3
| Macromolecule | Name: Replication factor C subunit 3 / type: protein_or_peptide / ID: 5 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 40.614332 KDa |
| Recombinant expression | Organism: Insect cell expression vector pTIE1 (others) |
| Sequence | String: MSLWVDKYRP CSLGRLDYHK EQAAQLRNLV QCGDFPHLLV YGPSGAGKKT RIMCILRELY GVGVEKLRIE HQTITTPSKK KIEISTIAS NYHLEVNPSD AGNSDRVVIQ EMLKTVAQSQ QLETNSQRDF KVVLLTEVDK LTKDAQHALR RTMEKYMSTC R LILCCNST ...String: MSLWVDKYRP CSLGRLDYHK EQAAQLRNLV QCGDFPHLLV YGPSGAGKKT RIMCILRELY GVGVEKLRIE HQTITTPSKK KIEISTIAS NYHLEVNPSD AGNSDRVVIQ EMLKTVAQSQ QLETNSQRDF KVVLLTEVDK LTKDAQHALR RTMEKYMSTC R LILCCNST SKVIPPIRSR CLAVRVPAPS IEDICHVLST VCKKEGLNLP SQLAHRLAEK SCRNLRKALL MCEACRVQQY PF TADQEIP ETDWEVYLRE TANAIVSQQT PQRLLEVRGR LYELLTHCIP PEIIMKGLLS ELLHNCDGQL KGEVAQMAAY YEH RLQLGS KAIYHLEAFV AKFMALYKKF MEDGLEGMMF UniProtKB:  Replication factor C subunit 3 Replication factor C subunit 3 |
-Macromolecule #6: MAGNESIUM ION
| Macromolecule | Name: MAGNESIUM ION / type: ligand / ID: 6 / Number of copies: 3 / Formula: MG |
|---|---|
| Molecular weight | Theoretical: 24.305 Da |
-Macromolecule #7: PHOSPHOTHIOPHOSPHORIC ACID-ADENYLATE ESTER
| Macromolecule | Name: PHOSPHOTHIOPHOSPHORIC ACID-ADENYLATE ESTER / type: ligand / ID: 7 / Number of copies: 3 / Formula: AGS |
|---|---|
| Molecular weight | Theoretical: 523.247 Da |
| Chemical component information |  ChemComp-AGS: |
-Macromolecule #8: ADENOSINE-5'-DIPHOSPHATE
| Macromolecule | Name: ADENOSINE-5'-DIPHOSPHATE / type: ligand / ID: 8 / Number of copies: 1 / Formula: ADP |
|---|---|
| Molecular weight | Theoretical: 427.201 Da |
| Chemical component information |  ChemComp-ADP: |
-Experimental details
-Structure determination
| Method |  cryo EM cryo EM |
|---|---|
 Processing Processing |  single particle reconstruction single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.3 mg/mL |
|---|---|
| Buffer | pH: 7.5 |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 280 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD Bright-field microscopy / Nominal defocus max: 1.9000000000000001 µm / Nominal defocus min: 1.2 µm Bright-field microscopy / Nominal defocus max: 1.9000000000000001 µm / Nominal defocus min: 1.2 µm |
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 60.0 e/Å2 |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
-Atomic model buiding 1
| Refinement | Space: REAL / Protocol: RIGID BODY FIT / Overall B value: 123 |
|---|---|
| Output model |  PDB-8unj: |
 Movie
Movie Controller
Controller











 Z
Z Y
Y X
X


































