[English] 日本語
 Yorodumi
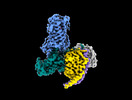
Yorodumi- EMDB-38401: Latanoprost acid bound Prostaglandin F2-alpha receptor-Gq Protein... -
+ Open data
Open data
- Basic information
Basic information
| Entry | 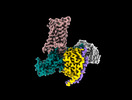 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Latanoprost acid bound Prostaglandin F2-alpha receptor-Gq Protein Complex | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords |  cryo-EM / cryo-EM /  Complex / Complex /  Signaling Protein / Signaling Protein /  MEMBRANE PROTEIN MEMBRANE PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology information prostaglandin F receptor activity / prostaglandin F receptor activity /  parturition / Prostanoid ligand receptors / cellular response to prostaglandin D stimulus / : / Olfactory Signaling Pathway / Activation of the phototransduction cascade / G beta:gamma signalling through PLC beta / Presynaptic function of Kainate receptors / Thromboxane signalling through TP receptor ... parturition / Prostanoid ligand receptors / cellular response to prostaglandin D stimulus / : / Olfactory Signaling Pathway / Activation of the phototransduction cascade / G beta:gamma signalling through PLC beta / Presynaptic function of Kainate receptors / Thromboxane signalling through TP receptor ... prostaglandin F receptor activity / prostaglandin F receptor activity /  parturition / Prostanoid ligand receptors / cellular response to prostaglandin D stimulus / : / Olfactory Signaling Pathway / Activation of the phototransduction cascade / G beta:gamma signalling through PLC beta / Presynaptic function of Kainate receptors / Thromboxane signalling through TP receptor / G-protein activation / G protein-coupled acetylcholine receptor signaling pathway / Activation of G protein gated Potassium channels / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / Prostacyclin signalling through prostacyclin receptor / Glucagon signaling in metabolic regulation / G beta:gamma signalling through CDC42 / adenylate cyclase-activating G protein-coupled receptor signaling pathway / ADP signalling through P2Y purinoceptor 12 / G beta:gamma signalling through BTK / Sensory perception of sweet, bitter, and umami (glutamate) taste / Synthesis, secretion, and inactivation of Glucagon-like Peptide-1 (GLP-1) / photoreceptor disc membrane / Adrenaline,noradrenaline inhibits insulin secretion / Glucagon-type ligand receptors / Vasopressin regulates renal water homeostasis via Aquaporins / G alpha (z) signalling events / cellular response to catecholamine stimulus / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / ADORA2B mediated anti-inflammatory cytokines production / adenylate cyclase-activating dopamine receptor signaling pathway / ADP signalling through P2Y purinoceptor 1 / G beta:gamma signalling through PI3Kgamma / cellular response to prostaglandin E stimulus / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / sensory perception of taste / GPER1 signaling / G-protein beta-subunit binding / parturition / Prostanoid ligand receptors / cellular response to prostaglandin D stimulus / : / Olfactory Signaling Pathway / Activation of the phototransduction cascade / G beta:gamma signalling through PLC beta / Presynaptic function of Kainate receptors / Thromboxane signalling through TP receptor / G-protein activation / G protein-coupled acetylcholine receptor signaling pathway / Activation of G protein gated Potassium channels / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / Prostacyclin signalling through prostacyclin receptor / Glucagon signaling in metabolic regulation / G beta:gamma signalling through CDC42 / adenylate cyclase-activating G protein-coupled receptor signaling pathway / ADP signalling through P2Y purinoceptor 12 / G beta:gamma signalling through BTK / Sensory perception of sweet, bitter, and umami (glutamate) taste / Synthesis, secretion, and inactivation of Glucagon-like Peptide-1 (GLP-1) / photoreceptor disc membrane / Adrenaline,noradrenaline inhibits insulin secretion / Glucagon-type ligand receptors / Vasopressin regulates renal water homeostasis via Aquaporins / G alpha (z) signalling events / cellular response to catecholamine stimulus / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / ADORA2B mediated anti-inflammatory cytokines production / adenylate cyclase-activating dopamine receptor signaling pathway / ADP signalling through P2Y purinoceptor 1 / G beta:gamma signalling through PI3Kgamma / cellular response to prostaglandin E stimulus / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / sensory perception of taste / GPER1 signaling / G-protein beta-subunit binding /  heterotrimeric G-protein complex / Inactivation, recovery and regulation of the phototransduction cascade / heterotrimeric G-protein complex / Inactivation, recovery and regulation of the phototransduction cascade /  extracellular vesicle / G alpha (12/13) signalling events / signaling receptor complex adaptor activity / Thrombin signalling through proteinase activated receptors (PARs) / response to estradiol / retina development in camera-type eye / extracellular vesicle / G alpha (12/13) signalling events / signaling receptor complex adaptor activity / Thrombin signalling through proteinase activated receptors (PARs) / response to estradiol / retina development in camera-type eye /  GTPase binding / Ca2+ pathway / phospholipase C-activating G protein-coupled receptor signaling pathway / positive regulation of cytosolic calcium ion concentration / G alpha (i) signalling events / fibroblast proliferation / G alpha (s) signalling events / G alpha (q) signalling events / Ras protein signal transduction / cell population proliferation / response to lipopolysaccharide / Extra-nuclear estrogen signaling / GTPase binding / Ca2+ pathway / phospholipase C-activating G protein-coupled receptor signaling pathway / positive regulation of cytosolic calcium ion concentration / G alpha (i) signalling events / fibroblast proliferation / G alpha (s) signalling events / G alpha (q) signalling events / Ras protein signal transduction / cell population proliferation / response to lipopolysaccharide / Extra-nuclear estrogen signaling /  inflammatory response / G protein-coupled receptor signaling pathway / lysosomal membrane / inflammatory response / G protein-coupled receptor signaling pathway / lysosomal membrane /  GTPase activity / GTPase activity /  synapse / positive regulation of cell population proliferation / protein-containing complex binding / positive regulation of gene expression / negative regulation of apoptotic process / synapse / positive regulation of cell population proliferation / protein-containing complex binding / positive regulation of gene expression / negative regulation of apoptotic process /  signal transduction / extracellular exosome / extracellular region / signal transduction / extracellular exosome / extracellular region /  membrane / membrane /  plasma membrane / plasma membrane /  cytosol / cytosol /  cytoplasm cytoplasmSimilarity search - Function | |||||||||
| Biological species |   Homo sapiens (human) / synthetic construct (others) Homo sapiens (human) / synthetic construct (others) | |||||||||
| Method |  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 2.85 Å cryo EM / Resolution: 2.85 Å | |||||||||
 Authors Authors | Zhang X / Li X / Liu G / Gong W | |||||||||
| Funding support |  China, 2 items China, 2 items
| |||||||||
 Citation Citation |  Journal: Cell Rep / Year: 2024 Journal: Cell Rep / Year: 2024Title: Structural basis for ligand recognition and activation of the prostanoid receptors. Authors: Xiu Li / Xuan Zhang / Xin Wen / Daolai Zhang / Changxiu Qu / Xinyi Miao / Wenkai Zhang / Ru Zhang / Guibing Liu / Peng Xiao / Jin-Peng Sun / Weimin Gong /  Abstract: Prostaglandin F (PGF) and thromboxane A (TXA) are endogenous arachidonic acid metabolites, modulating diverse physiological processes including inflammation and cardiovascular homeostasis through ...Prostaglandin F (PGF) and thromboxane A (TXA) are endogenous arachidonic acid metabolites, modulating diverse physiological processes including inflammation and cardiovascular homeostasis through activating PGF receptor (FP) and TXA receptor (TP). Ligands targeting FP and TP have demonstrated efficacy in treating conditions like glaucoma and cardiovascular diseases in humans, as well as reproductive-related diseases in animals. Here, we present five cryoelectron microscopy structures illustrating FP and TP in complex with G and bound to PGF (endogenous ligand), latanoprost acid (a clinical drug), and two other synthetic agonists. Combined with mutational and functional studies, these structures reveal not only structural features for the specific recognition of endogenous ligands and attainment of receptor selectivity of FP and TP but also the common mechanisms of receptor activation and G protein coupling. The findings may enrich our knowledge of ligand recognition and signal transduction of the prostanoid receptor family and facilitate rational ligand design toward these two receptors. | |||||||||
| History |
|
- Structure visualization
Structure visualization
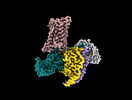
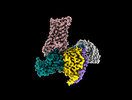
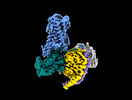
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_38401.map.gz emd_38401.map.gz | 18.2 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-38401-v30.xml emd-38401-v30.xml emd-38401.xml emd-38401.xml | 19.3 KB 19.3 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_38401.png emd_38401.png | 48.2 KB | ||
| Filedesc metadata |  emd-38401.cif.gz emd-38401.cif.gz | 6.6 KB | ||
| Others |  emd_38401_half_map_1.map.gz emd_38401_half_map_1.map.gz emd_38401_half_map_2.map.gz emd_38401_half_map_2.map.gz | 20.7 MB 20.7 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-38401 http://ftp.pdbj.org/pub/emdb/structures/EMD-38401 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-38401 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-38401 | HTTPS FTP |
-Related structure data
| Related structure data |  8xjmMC  8xjkC  8xjlC  8xjnC  8xjoC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_38401.map.gz / Format: CCP4 / Size: 27 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_38401.map.gz / Format: CCP4 / Size: 27 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 1.07 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #1
| File | emd_38401_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #2
| File | emd_38401_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Latanoprost acid-FP-miniGq-scFv16 complex
| Entire | Name: Latanoprost acid-FP-miniGq-scFv16 complex |
|---|---|
| Components |
|
-Supramolecule #1: Latanoprost acid-FP-miniGq-scFv16 complex
| Supramolecule | Name: Latanoprost acid-FP-miniGq-scFv16 complex / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#5 |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Engineered miniGq
| Macromolecule | Name: Engineered miniGq / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
| Molecular weight | Theoretical: 28.084832 KDa |
| Recombinant expression | Organism:   Spodoptera frugiperda (fall armyworm) Spodoptera frugiperda (fall armyworm) |
| Sequence | String: MGSTVSAEDK AAAERSKMID KNLREDGEKA RRTLRLLLLG ADNSGKSTIV KQMRILHGGS GGSGGTSGIF ETKFQVDKVN FHMFDVGGQ RDERRKWIQC FNDVTAIIFV VDSSDYNRLQ EALNDFKSIW NNRWLRTISV ILFLNKQDLL AEKVLAGKSK I EDYFPEFA ...String: MGSTVSAEDK AAAERSKMID KNLREDGEKA RRTLRLLLLG ADNSGKSTIV KQMRILHGGS GGSGGTSGIF ETKFQVDKVN FHMFDVGGQ RDERRKWIQC FNDVTAIIFV VDSSDYNRLQ EALNDFKSIW NNRWLRTISV ILFLNKQDLL AEKVLAGKSK I EDYFPEFA RYTTPEDATP EPGEDPRVTR AKYFIRKEFV DISTASGDGR HICYPHFTCA VDTENARRIF NDCKDIILQM NL REYNLV |
-Macromolecule #2: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 40.845559 KDa |
| Recombinant expression | Organism:   Spodoptera frugiperda (fall armyworm) Spodoptera frugiperda (fall armyworm) |
| Sequence | String: MHHHHHHGSS GSELDQLRQE AEQLKNQIRD ARKACADATL SQITNNIDPV GRIQMRTRRT LRGHLAKIYA MHWGTDSRLL VSASQDGKL IIWDSYTTNK VHAIPLRSSW VMTCAYAPSG NYVACGGLDN ICSIYNLKTR EGNVRVSREL AGHTGYLSCC R FLDDNQIV ...String: MHHHHHHGSS GSELDQLRQE AEQLKNQIRD ARKACADATL SQITNNIDPV GRIQMRTRRT LRGHLAKIYA MHWGTDSRLL VSASQDGKL IIWDSYTTNK VHAIPLRSSW VMTCAYAPSG NYVACGGLDN ICSIYNLKTR EGNVRVSREL AGHTGYLSCC R FLDDNQIV TSSGDTTCAL WDIETGQQTT TFTGHTGDVM SLSLAPDTRL FVSGACDASA KLWDVREGMC RQTFTGHESD IN AICFFPN GNAFATGSDD ATCRLFDLRA DQELMTYSHD NIICGITSVS FSKSGRLLLA GYDDFNCNVW DALKADRAGV LAG HDNRVS CLGVTDDGMA VATGSWDSFL KIWNGSSGGG GSGGGGSSGV SGWRLFKKIS UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 |
-Macromolecule #3: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 7.861143 KDa |
| Recombinant expression | Organism:   Spodoptera frugiperda (fall armyworm) Spodoptera frugiperda (fall armyworm) |
| Sequence | String: MASNNTASIA QARKLVEQLK MEANIDRIKV SKAAADLMAY CEAHAKEDPL LTPVPASENP FREKKFFCAI L UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 |
-Macromolecule #4: Antibody fragment scFv16
| Macromolecule | Name: Antibody fragment scFv16 / type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
| Molecular weight | Theoretical: 27.259344 KDa |
| Recombinant expression | Organism:   Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: VQLVESGGGL VQPGGSRKLS CSASGFAFSS FGMHWVRQAP EKGLEWVAYI SSGSGTIYYA DTVKGRFTIS RDDPKNTLFL QMTSLRSED TAMYYCVRSI YYYGSSPFDF WGQGTTLTVS SGGGGSGGGG SGGGGSDIVM TQATSSVPVT PGESVSISCR S SKSLLHSN ...String: VQLVESGGGL VQPGGSRKLS CSASGFAFSS FGMHWVRQAP EKGLEWVAYI SSGSGTIYYA DTVKGRFTIS RDDPKNTLFL QMTSLRSED TAMYYCVRSI YYYGSSPFDF WGQGTTLTVS SGGGGSGGGG SGGGGSDIVM TQATSSVPVT PGESVSISCR S SKSLLHSN GNTYLYWFLQ RPGQSPQLLI YRMSNLASGV PDRFSGSGSG TAFTLTISRL EAEDVGVYYC MQHLEYPLTF GA GTKLELL EENLYFQ |
-Macromolecule #5: Fusion tag,Prostaglandin F2-alpha receptor,LgBiT
| Macromolecule | Name: Fusion tag,Prostaglandin F2-alpha receptor,LgBiT / type: protein_or_peptide / ID: 5 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
| Molecular weight | Theoretical: 64.767266 KDa |
| Recombinant expression | Organism:   Spodoptera frugiperda (fall armyworm) Spodoptera frugiperda (fall armyworm) |
| Sequence | String: DYKDDDDHHH HHHHHGQPGN GSAFLLAPNG SHAPDHNVTQ QRDEGGSGQP GNGSAFLLAP NGSHAPDHNV TQQRDEENLY FQGVDMSMN NSKQLVSPAA ALLSNTTCQT ENRLSVFFSV IFMTVGILSN SLAIAILMKA YQRFRQKSKA SFLLLASGLV I TDFFGHLI ...String: DYKDDDDHHH HHHHHGQPGN GSAFLLAPNG SHAPDHNVTQ QRDEGGSGQP GNGSAFLLAP NGSHAPDHNV TQQRDEENLY FQGVDMSMN NSKQLVSPAA ALLSNTTCQT ENRLSVFFSV IFMTVGILSN SLAIAILMKA YQRFRQKSKA SFLLLASGLV I TDFFGHLI NGAIAVFVYA SDKEWIRFDQ SNVLCSIFGI CMVFSGLCPL LLGSVMAIER CIGVTKPIFH STKITSKHVK MM LSGVCLF AVFIALLPIL GHRDYKIQAS RTWCFYNTED IKDWEDRFYL LLFSFLGLLA LGVSLLCNAI TGITLLRVKF KSQ QHRQGR SHHLEMVIQL LAIMCVSCIC WSPFLVTMAN IGINGNHSLE TCETTLFALR MATWNQILDP WVYILLRKAV LKNL YKLAS QCCGVHVGSS GGGGSGGGGS SGAAAVFTLE DFVGDWEQTA AYNLDQVLEQ GGVSSLLQNL AVSVTPIQRI VRSGE NALK IDIHVIIPYE GLSADQMAQI EEVFKVVYPV DDHHFKVILP YGTLVIDGVT PNMLNYFGRP YEGIAVFDGK KITVTG TLW NGNKIIDERL ITPDGSMLFR VTINS UniProtKB: Prostaglandin F2-alpha receptor |
-Macromolecule #6: Z-7-[(1R,2R,3R,5S)-3,5-bis(oxidanyl)-2-[(3R)-3-oxidanyl-5-phenyl-...
| Macromolecule | Name: Z-7-[(1R,2R,3R,5S)-3,5-bis(oxidanyl)-2-[(3R)-3-oxidanyl-5-phenyl-pentyl]cyclopentyl]hept-5-enoic acid type: ligand / ID: 6 / Number of copies: 1 / Formula: 7WT |
|---|---|
| Molecular weight | Theoretical: 390.513 Da |
| Chemical component information |  ChemComp-7WT: |
-Experimental details
-Structure determination
| Method |  cryo EM cryo EM |
|---|---|
 Processing Processing |  single particle reconstruction single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD Bright-field microscopy / Nominal defocus max: 2.2 µm / Nominal defocus min: 1.2 µm Bright-field microscopy / Nominal defocus max: 2.2 µm / Nominal defocus min: 1.2 µm |
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 55.0 e/Å2 |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: INSILICO MODEL |
|---|---|
| Initial angle assignment | Type: PROJECTION MATCHING |
| Final angle assignment | Type: PROJECTION MATCHING |
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 2.85 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 91516 |
 Movie
Movie Controller
Controller
























 Z
Z Y
Y X
X

















