[English] 日本語
 Yorodumi
Yorodumi- EMDB-37712: Small-heat shock protein from Methanocaldococcus jannaschii, Hsp16.5 -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Small-heat shock protein from Methanocaldococcus jannaschii, Hsp16.5 | ||||||||||||
 Map data Map data | |||||||||||||
 Sample Sample |
| ||||||||||||
 Keywords Keywords | Hsp16.5 / molecular chaperone / oligomeric protein / small heat-shock protein / stress response / CHAPERONE | ||||||||||||
| Biological species |   Methanocaldococcus jannaschii DSM 2661 (archaea) Methanocaldococcus jannaschii DSM 2661 (archaea) | ||||||||||||
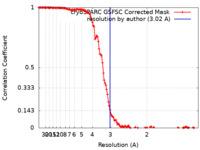
| Method | single particle reconstruction / cryo EM / Resolution: 3.02 Å | ||||||||||||
 Authors Authors | Lee J / Ryu B / Kim T / Kim KK | ||||||||||||
| Funding support |  Korea, Republic Of, 3 items Korea, Republic Of, 3 items
| ||||||||||||
 Citation Citation |  Journal: Int J Biol Macromol / Year: 2024 Journal: Int J Biol Macromol / Year: 2024Title: Cryo-EM structure of a 16.5-kDa small heat-shock protein from Methanocaldococcus jannaschii. Authors: Joohyun Lee / Bumhan Ryu / Truc Kim / Kyeong Kyu Kim /  Abstract: The small heat-shock protein (sHSP) from the archaea Methanocaldococcus jannaschii, MjsHSP16.5, functions as a broad substrate ATP-independent holding chaperone protecting misfolded proteins from ...The small heat-shock protein (sHSP) from the archaea Methanocaldococcus jannaschii, MjsHSP16.5, functions as a broad substrate ATP-independent holding chaperone protecting misfolded proteins from aggregation under stress conditions. This protein is the first sHSP characterized by X-ray crystallography, thereby contributing significantly to our understanding of sHSPs. However, despite numerous studies assessing its functions and structures, the precise arrangement of the N-terminal domains (NTDs) within this sHSP cage remains elusive. Here we present the cryo-electron microscopy (cryo-EM) structure of MjsHSP16.5 at 2.49-Å resolution. The subunits of MjsHSP16.5 in the cryo-EM structure exhibit lesser compaction compared to their counterparts in the crystal structure. This structural feature holds particular significance in relation to the biophysical properties of MjsHSP16.5, suggesting a close resemblance to this sHSP native state. Additionally, our cryo-EM structure unveils the density of residues 24-33 within the NTD of MjsHSP16.5, a feature that typically remains invisible in the majority of its crystal structures. Notably, these residues show a propensity to adopt a β-strand conformation and engage in antiparallel interactions with strand β1, both intra- and inter-subunit modes. These structural insights are corroborated by structural predictions, disulfide bond cross-linking studies of Cys-substitution mutants, and protein disaggregation assays. A comprehensive understanding of the structural features of MjsHSP16.5 expectedly holds the potential to inspire a wide range of interdisciplinary applications, owing to the renowned versatility of this sHSP as a nanoscale protein platform. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_37712.map.gz emd_37712.map.gz | 107.8 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-37712-v30.xml emd-37712-v30.xml emd-37712.xml emd-37712.xml | 16.4 KB 16.4 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_37712_fsc.xml emd_37712_fsc.xml | 14.4 KB | Display |  FSC data file FSC data file |
| Images |  emd_37712.png emd_37712.png | 161.2 KB | ||
| Masks |  emd_37712_msk_1.map emd_37712_msk_1.map | 216 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-37712.cif.gz emd-37712.cif.gz | 4.1 KB | ||
| Others |  emd_37712_additional_1.map.gz emd_37712_additional_1.map.gz emd_37712_half_map_1.map.gz emd_37712_half_map_1.map.gz emd_37712_half_map_2.map.gz emd_37712_half_map_2.map.gz | 171.1 MB 170.7 MB 170.7 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-37712 http://ftp.pdbj.org/pub/emdb/structures/EMD-37712 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-37712 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-37712 | HTTPS FTP |
-Validation report
| Summary document |  emd_37712_validation.pdf.gz emd_37712_validation.pdf.gz | 980.8 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_37712_full_validation.pdf.gz emd_37712_full_validation.pdf.gz | 980.4 KB | Display | |
| Data in XML |  emd_37712_validation.xml.gz emd_37712_validation.xml.gz | 21.7 KB | Display | |
| Data in CIF |  emd_37712_validation.cif.gz emd_37712_validation.cif.gz | 27.9 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-37712 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-37712 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-37712 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-37712 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_37712.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_37712.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 0.673 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_37712_msk_1.map emd_37712_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
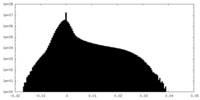
| Projections & Slices |
| ||||||||||||
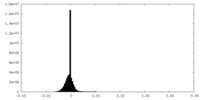
| Density Histograms |
-Additional map: #1
| File | emd_37712_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
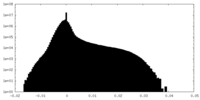
| Projections & Slices |
| ||||||||||||
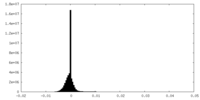
| Density Histograms |
-Half map: #2
| File | emd_37712_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_37712_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Small-heat shock protein from Methanocaldococcus jannaschii, Hsp16.5
| Entire | Name: Small-heat shock protein from Methanocaldococcus jannaschii, Hsp16.5 |
|---|---|
| Components |
|
-Supramolecule #1: Small-heat shock protein from Methanocaldococcus jannaschii, Hsp16.5
| Supramolecule | Name: Small-heat shock protein from Methanocaldococcus jannaschii, Hsp16.5 type: complex / ID: 1 / Parent: 0 |
|---|---|
| Source (natural) | Organism:   Methanocaldococcus jannaschii DSM 2661 (archaea) Methanocaldococcus jannaschii DSM 2661 (archaea) |
| Molecular weight | Theoretical: 396 KDa |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 1.7 mg/mL |
|---|---|
| Buffer | pH: 7.2 |
| Vitrification | Cryogen name: ETHANE / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON III (4k x 4k) / Detector mode: COUNTING / Average electron dose: 30.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.3000000000000003 µm / Nominal defocus min: 0.8 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Protocol: AB INITIO MODEL |
|---|
 Movie
Movie Controller
Controller





 Z
Z Y
Y X
X