[English] 日本語
 Yorodumi
Yorodumi- EMDB-37251: Structure of the human ATP synthase bound to bedaquiline (composite) -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of the human ATP synthase bound to bedaquiline (composite) | ||||||||||||
 Map data Map data | |||||||||||||
 Sample Sample |
| ||||||||||||
 Keywords Keywords |  ATP synthase / ATP synthase /  Human / Human /  cryo-EM / cryo-EM /  Membrane protein Membrane protein | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationmitochondrial proton-transporting ATP synthase complex binding / regulation of ATP metabolic process / negative regulation of cell adhesion involved in substrate-bound cell migration / regulation of protein targeting to mitochondrion / Formation of ATP by chemiosmotic coupling / Cristae formation / positive regulation of proteolysis involved in protein catabolic process / positive regulation of autophagy of mitochondrion in response to mitochondrial depolarization / ATP biosynthetic process /  angiostatin binding ...mitochondrial proton-transporting ATP synthase complex binding / regulation of ATP metabolic process / negative regulation of cell adhesion involved in substrate-bound cell migration / regulation of protein targeting to mitochondrion / Formation of ATP by chemiosmotic coupling / Cristae formation / positive regulation of proteolysis involved in protein catabolic process / positive regulation of autophagy of mitochondrion in response to mitochondrial depolarization / ATP biosynthetic process / angiostatin binding ...mitochondrial proton-transporting ATP synthase complex binding / regulation of ATP metabolic process / negative regulation of cell adhesion involved in substrate-bound cell migration / regulation of protein targeting to mitochondrion / Formation of ATP by chemiosmotic coupling / Cristae formation / positive regulation of proteolysis involved in protein catabolic process / positive regulation of autophagy of mitochondrion in response to mitochondrial depolarization / ATP biosynthetic process /  angiostatin binding / ATPase inhibitor activity / mitochondrial depolarization / positive regulation of mitochondrial outer membrane permeabilization involved in apoptotic signaling pathway / angiostatin binding / ATPase inhibitor activity / mitochondrial depolarization / positive regulation of mitochondrial outer membrane permeabilization involved in apoptotic signaling pathway /  Mitochondrial protein import / negative regulation of ATP-dependent activity / mitochondrial proton-transporting ATP synthase complex assembly / negative regulation of hydrolase activity / mitochondrial proton-transporting ATP synthase, catalytic core / mitochondrial proton-transporting ATP synthase, stator stalk / Mitochondrial protein import / negative regulation of ATP-dependent activity / mitochondrial proton-transporting ATP synthase complex assembly / negative regulation of hydrolase activity / mitochondrial proton-transporting ATP synthase, catalytic core / mitochondrial proton-transporting ATP synthase, stator stalk /  enzyme inhibitor activity / proton-transporting ATP synthase complex / cellular response to interleukin-7 / enzyme inhibitor activity / proton-transporting ATP synthase complex / cellular response to interleukin-7 /  oxidative phosphorylation / mitochondrial proton-transporting ATP synthase complex, coupling factor F(o) / response to muscle activity / response to copper ion / heme biosynthetic process / mitochondrial proton-transporting ATP synthase complex / mitochondrial proton-transporting ATP synthase complex, catalytic sector F(1) / mitochondrial nucleoid / proton motive force-driven mitochondrial ATP synthesis / negative regulation of endothelial cell proliferation / proton-transporting ATP synthase complex, coupling factor F(o) / proton motive force-driven ATP synthesis / proton transmembrane transporter activity / cellular response to nitric oxide / response to hyperoxia / proton-transporting ATP synthase complex, catalytic core F(1) / positive regulation of blood vessel endothelial cell migration / MHC class I protein binding / oxidative phosphorylation / mitochondrial proton-transporting ATP synthase complex, coupling factor F(o) / response to muscle activity / response to copper ion / heme biosynthetic process / mitochondrial proton-transporting ATP synthase complex / mitochondrial proton-transporting ATP synthase complex, catalytic sector F(1) / mitochondrial nucleoid / proton motive force-driven mitochondrial ATP synthesis / negative regulation of endothelial cell proliferation / proton-transporting ATP synthase complex, coupling factor F(o) / proton motive force-driven ATP synthesis / proton transmembrane transporter activity / cellular response to nitric oxide / response to hyperoxia / proton-transporting ATP synthase complex, catalytic core F(1) / positive regulation of blood vessel endothelial cell migration / MHC class I protein binding /  aerobic respiration / aerobic respiration /  H+-transporting two-sector ATPase / substantia nigra development / proton-transporting ATPase activity, rotational mechanism / reactive oxygen species metabolic process / proton transmembrane transport / proton-transporting ATP synthase activity, rotational mechanism / cellular response to dexamethasone stimulus / H+-transporting two-sector ATPase / substantia nigra development / proton-transporting ATPase activity, rotational mechanism / reactive oxygen species metabolic process / proton transmembrane transport / proton-transporting ATP synthase activity, rotational mechanism / cellular response to dexamethasone stimulus /  erythrocyte differentiation / generation of precursor metabolites and energy / erythrocyte differentiation / generation of precursor metabolites and energy /  ADP binding / ADP binding /  regulation of intracellular pH / regulation of intracellular pH /  mitochondrial membrane / Transcriptional activation of mitochondrial biogenesis / lipid metabolic process / osteoblast differentiation / mitochondrial membrane / Transcriptional activation of mitochondrial biogenesis / lipid metabolic process / osteoblast differentiation /  ATPase binding / ATPase binding /  angiogenesis / response to ethanol / angiogenesis / response to ethanol /  nuclear membrane / nuclear membrane /  mitochondrial inner membrane / mitochondrial inner membrane /  protease binding / protease binding /  calmodulin binding / calmodulin binding /  hydrolase activity / hydrolase activity /  mitochondrial matrix / mitochondrial matrix /  membrane raft / membrane raft /  lipid binding / lipid binding /  enzyme binding / enzyme binding /  cell surface / cell surface /  ATP hydrolysis activity / protein-containing complex / ATP hydrolysis activity / protein-containing complex /  mitochondrion / mitochondrion /  RNA binding / extracellular exosome / RNA binding / extracellular exosome /  ATP binding / ATP binding /  membrane / identical protein binding / membrane / identical protein binding /  nucleus / nucleus /  plasma membrane plasma membraneSimilarity search - Function | ||||||||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | ||||||||||||
| Method |  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 2.89 Å cryo EM / Resolution: 2.89 Å | ||||||||||||
 Authors Authors | Lai Y / Zhang Y / Gong H | ||||||||||||
| Funding support |  China, 3 items China, 3 items
| ||||||||||||
 Citation Citation |  Journal: To Be Published Journal: To Be PublishedTitle: Structure of Mycobacterium tuberculosis ATP synthase Authors: Zhang Y / Lai Y / Liu F / Rao Z / Gong H | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_37251.map.gz emd_37251.map.gz | 468.4 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-37251-v30.xml emd-37251-v30.xml emd-37251.xml emd-37251.xml | 27.1 KB 27.1 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_37251.png emd_37251.png | 41.8 KB | ||
| Filedesc metadata |  emd-37251.cif.gz emd-37251.cif.gz | 7.6 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-37251 http://ftp.pdbj.org/pub/emdb/structures/EMD-37251 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-37251 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-37251 | HTTPS FTP |
-Related structure data
| Related structure data |  8ki3MC 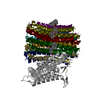 8j57C  8j58C  8jr0C  8jr1C  8khfC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_37251.map.gz / Format: CCP4 / Size: 512 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_37251.map.gz / Format: CCP4 / Size: 512 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 0.73 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
- Sample components
Sample components
+Entire : human ATP synthase
+Supramolecule #1: human ATP synthase
+Macromolecule #1: ATP synthase subunit alpha, mitochondrial
+Macromolecule #2: ATP synthase subunit beta, mitochondrial
+Macromolecule #3: ATP synthase subunit gamma, mitochondrial
+Macromolecule #4: ATPase inhibitor, mitochondrial
+Macromolecule #5: ATP synthase subunit O, mitochondrial
+Macromolecule #6: ATP synthase F(0) complex subunit C1, mitochondrial
+Macromolecule #7: ATP synthase subunit delta, mitochondrial
+Macromolecule #8: ATP synthase subunit epsilon, mitochondrial
+Macromolecule #9: ATP synthase F(0) complex subunit B1, mitochondrial
+Macromolecule #10: ATP synthase subunit d, mitochondrial
+Macromolecule #11: ATP synthase subunit a
+Macromolecule #12: ATP synthase subunit ATP5MJ, mitochondrial
+Macromolecule #13: ATP synthase protein 8
+Macromolecule #14: ATP synthase subunit f, mitochondrial
+Macromolecule #15: ATP synthase subunit g, mitochondrial
+Macromolecule #16: ATP synthase subunit e, mitochondrial
+Macromolecule #17: ATP synthase-coupling factor 6, mitochondrial
+Macromolecule #18: ADENOSINE-5'-TRIPHOSPHATE
+Macromolecule #19: MAGNESIUM ION
+Macromolecule #20: ADENOSINE-5'-DIPHOSPHATE
+Macromolecule #21: Bedaquiline
-Experimental details
-Structure determination
| Method |  cryo EM cryo EM |
|---|---|
 Processing Processing |  single particle reconstruction single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD Bright-field microscopy / Nominal defocus max: 2.4 µm / Nominal defocus min: 1.2 µm Bright-field microscopy / Nominal defocus max: 2.4 µm / Nominal defocus min: 1.2 µm |
| Image recording | Film or detector model: FEI FALCON IV (4k x 4k) / Average electron dose: 50.0 e/Å2 |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: PDB ENTRY PDB model - PDB ID: |
|---|---|
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 2.89 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 84037 |
 Movie
Movie Controller
Controller