[English] 日本語
 Yorodumi
Yorodumi- EMDB-32760: Cryo-EM structure of human glucose transporter GLUT4 bound to cyt... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of human glucose transporter GLUT4 bound to cytochalasin B in lipid nanodiscs | |||||||||
 Map data Map data | Cryo-EM structure of human glucose transporter GLUT4 | |||||||||
 Sample Sample |
| |||||||||
| Function / homology |  Function and homology information Function and homology informationhexose transmembrane transporter activity / glucose uniporter activity / monosaccharide transmembrane transport / regulation of synaptic vesicle budding from presynaptic endocytic zone membrane / amylopectin biosynthetic process / positive regulation of brain-derived neurotrophic factor receptor signaling pathway / glucose transmembrane transporter activity / D-glucose transmembrane transporter activity / Cellular hexose transport / glucose import in response to insulin stimulus ...hexose transmembrane transporter activity / glucose uniporter activity / monosaccharide transmembrane transport / regulation of synaptic vesicle budding from presynaptic endocytic zone membrane / amylopectin biosynthetic process / positive regulation of brain-derived neurotrophic factor receptor signaling pathway / glucose transmembrane transporter activity / D-glucose transmembrane transporter activity / Cellular hexose transport / glucose import in response to insulin stimulus / insulin-responsive compartment / glucose transmembrane transport /  short-term memory / trans-Golgi network transport vesicle / vesicle membrane / cellular response to osmotic stress / short-term memory / trans-Golgi network transport vesicle / vesicle membrane / cellular response to osmotic stress /  clathrin-coated vesicle / glucose import / plasma membrane => GO:0005886 / transport across blood-brain barrier / clathrin-coated vesicle / glucose import / plasma membrane => GO:0005886 / transport across blood-brain barrier /  endomembrane system / endomembrane system /  long-term memory / brown fat cell differentiation / long-term memory / brown fat cell differentiation /  clathrin-coated pit / clathrin-coated pit /  T-tubule / T-tubule /  multivesicular body / multivesicular body /  sarcoplasmic reticulum / Translocation of SLC2A4 (GLUT4) to the plasma membrane / sarcoplasmic reticulum / Translocation of SLC2A4 (GLUT4) to the plasma membrane /  trans-Golgi network / cytoplasmic vesicle membrane / trans-Golgi network / cytoplasmic vesicle membrane /  sarcolemma / Transcriptional regulation of white adipocyte differentiation / cellular response to insulin stimulus / presynapse / sarcolemma / Transcriptional regulation of white adipocyte differentiation / cellular response to insulin stimulus / presynapse /  glucose homeostasis / cellular response to tumor necrosis factor / cellular response to hypoxia / response to ethanol / learning or memory / carbohydrate metabolic process / glucose homeostasis / cellular response to tumor necrosis factor / cellular response to hypoxia / response to ethanol / learning or memory / carbohydrate metabolic process /  membrane raft / external side of plasma membrane / perinuclear region of cytoplasm / extracellular exosome / membrane raft / external side of plasma membrane / perinuclear region of cytoplasm / extracellular exosome /  membrane / membrane /  plasma membrane / plasma membrane /  cytosol cytosolSimilarity search - Function | |||||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method |  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 3.25 Å cryo EM / Resolution: 3.25 Å | |||||||||
 Authors Authors | Yuan Y / Kong F / Xu H / Zhu A / Yan N / Yan C | |||||||||
| Funding support |  China, 2 items China, 2 items
| |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2022 Journal: Nat Commun / Year: 2022Title: Cryo-EM structure of human glucose transporter GLUT4. Authors: Yafei Yuan / Fang Kong / Hanwen Xu / Angqi Zhu / Nieng Yan / Chuangye Yan /   Abstract: GLUT4 is the primary glucose transporter in adipose and skeletal muscle tissues. Its cellular trafficking is regulated by insulin signaling. Failed or reduced plasma membrane localization of GLUT4 is ...GLUT4 is the primary glucose transporter in adipose and skeletal muscle tissues. Its cellular trafficking is regulated by insulin signaling. Failed or reduced plasma membrane localization of GLUT4 is associated with diabetes. Here, we report the cryo-EM structures of human GLUT4 bound to a small molecule inhibitor cytochalasin B (CCB) at resolutions of 3.3 Å in both detergent micelles and lipid nanodiscs. CCB-bound GLUT4 exhibits an inward-open conformation. Despite the nearly identical conformation of the transmembrane domain to GLUT1, the cryo-EM structure reveals an extracellular glycosylation site and an intracellular helix that is invisible in the crystal structure of GLUT1. The structural study presented here lays the foundation for further mechanistic investigation of the modulation of GLUT4 trafficking. Our methods for cryo-EM analysis of GLUT4 will also facilitate structural determination of many other small size solute carriers. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_32760.map.gz emd_32760.map.gz | 25.5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-32760-v30.xml emd-32760-v30.xml emd-32760.xml emd-32760.xml | 11.2 KB 11.2 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_32760.png emd_32760.png | 146.9 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-32760 http://ftp.pdbj.org/pub/emdb/structures/EMD-32760 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-32760 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-32760 | HTTPS FTP |
-Related structure data
| Related structure data |  7wsmMC  7wsnC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_32760.map.gz / Format: CCP4 / Size: 27 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_32760.map.gz / Format: CCP4 / Size: 27 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Cryo-EM structure of human glucose transporter GLUT4 | ||||||||||||||||||||
| Voxel size | X=Y=Z: 1.0825 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
- Sample components
Sample components
-Entire : GLUT4
| Entire | Name: GLUT4 |
|---|---|
| Components |
|
-Supramolecule #1: GLUT4
| Supramolecule | Name: GLUT4 / type: complex / Chimera: Yes / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Recombinant expression | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 56 kDa/nm |
-Macromolecule #1: Solute carrier family 2, facilitated glucose transporter member 4
| Macromolecule | Name: Solute carrier family 2, facilitated glucose transporter member 4 type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 56.108199 KDa |
| Recombinant expression | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MDYKDDDDKG TMPSGFQQIG SEDGEPPQQR VTGTLVLAVF SAVLGSLQFG YNIGVINAPQ KVIEQSYNET WLGRQGPEGP SSIPPGTLT TLWALSVAIF SVGGMISSFL IGIISQWLGR KRAMLVNNVL AVLGGSLMGL ANAAASYEML ILGRFLIGAY S GLTSGLVP ...String: MDYKDDDDKG TMPSGFQQIG SEDGEPPQQR VTGTLVLAVF SAVLGSLQFG YNIGVINAPQ KVIEQSYNET WLGRQGPEGP SSIPPGTLT TLWALSVAIF SVGGMISSFL IGIISQWLGR KRAMLVNNVL AVLGGSLMGL ANAAASYEML ILGRFLIGAY S GLTSGLVP MYVGEIAPTH LRGALGTLNQ LAIVIGILIA QVLGLESLLG TASLWPLLLG LTVLPALLQL VLLPFCPESP RY LYIIQNL EGPARKSLKR LTGWADVSGV LAELKDEKRK LERERPLSLL QLLGSRTHRQ PLIIAVVLQL SQQLSGINAV FYY STSIFE TAGVGQPAYA TIGAGVVNTV FTLVSVLLVE RAGRRTLHLL GLAGMCGCAI LMTVALLLLE RVPAMSYVSI VAIF GFVAF FEIGPGPIPW FIVAELFSQG PRPAAMAVAG FSNWTSNFII GMGFQYVAEA MGPYVFLLFA VLLLGFFIFT FLRVP ETRG RTFDQISAAF HRTPSLLEQE VKPSTELEYL GPDEND |
-Macromolecule #3: Cytochalasin B
| Macromolecule | Name: Cytochalasin B / type: ligand / ID: 3 / Number of copies: 1 / Formula: 5RH |
|---|---|
| Molecular weight | Theoretical: 479.608 Da |
| Chemical component information | 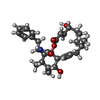 ChemComp-5RH: |
-Experimental details
-Structure determination
| Method |  cryo EM cryo EM |
|---|---|
 Processing Processing |  single particle reconstruction single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 1.8 mg/mL |
|---|---|
| Buffer | pH: 8 |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD Bright-field microscopy / Nominal defocus max: 2.5 µm / Nominal defocus min: 0.8 µm Bright-field microscopy / Nominal defocus max: 2.5 µm / Nominal defocus min: 0.8 µm |
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 50.0 e/Å2 |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD / Software - Name: cryoSPARC |
|---|---|
| Final angle assignment | Type: MAXIMUM LIKELIHOOD / Software - Name: cryoSPARC |
| Final reconstruction | Applied symmetry - Point group: C1 (asymmetric) / Resolution.type: BY AUTHOR / Resolution: 3.25 Å / Resolution method: FSC 0.143 CUT-OFF / Software - Name: cryoSPARC / Number images used: 73000 |
 Movie
Movie Controller
Controller












