[English] 日本語
 Yorodumi
Yorodumi- EMDB-32331: Cryo-EM structure of the alpha2A adrenergic receptor GoA signalin... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of the alpha2A adrenergic receptor GoA signaling complex bound to a G protein biased agonist | |||||||||
 Map data Map data | Cryo-EM structure of the alpha2A adrenergic receptor GoA signaling complex bound to a G protein biased agonist | |||||||||
 Sample Sample |
| |||||||||
| Function / homology |  Function and homology information Function and homology informationnegative regulation of uterine smooth muscle contraction / adenylate cyclase-inhibiting adrenergic receptor signaling pathway / phospholipase C-activating adrenergic receptor signaling pathway /  alpha2-adrenergic receptor activity / Adrenaline signalling through Alpha-2 adrenergic receptor / alpha2-adrenergic receptor activity / Adrenaline signalling through Alpha-2 adrenergic receptor /  alpha-2C adrenergic receptor binding / receptor transactivation / alpha-2C adrenergic receptor binding / receptor transactivation /  epinephrine binding / epinephrine binding /  alpha-1B adrenergic receptor binding / negative regulation of norepinephrine secretion ...negative regulation of uterine smooth muscle contraction / adenylate cyclase-inhibiting adrenergic receptor signaling pathway / phospholipase C-activating adrenergic receptor signaling pathway / alpha-1B adrenergic receptor binding / negative regulation of norepinephrine secretion ...negative regulation of uterine smooth muscle contraction / adenylate cyclase-inhibiting adrenergic receptor signaling pathway / phospholipase C-activating adrenergic receptor signaling pathway /  alpha2-adrenergic receptor activity / Adrenaline signalling through Alpha-2 adrenergic receptor / alpha2-adrenergic receptor activity / Adrenaline signalling through Alpha-2 adrenergic receptor /  alpha-2C adrenergic receptor binding / receptor transactivation / alpha-2C adrenergic receptor binding / receptor transactivation /  epinephrine binding / epinephrine binding /  alpha-1B adrenergic receptor binding / negative regulation of norepinephrine secretion / negative regulation of calcium ion transmembrane transporter activity / negative regulation of epinephrine secretion / alpha-1B adrenergic receptor binding / negative regulation of norepinephrine secretion / negative regulation of calcium ion transmembrane transporter activity / negative regulation of epinephrine secretion /  heterotrimeric G-protein binding / dopaminergic synapse / negative regulation of calcium ion-dependent exocytosis / mu-type opioid receptor binding / heterotrimeric G-protein binding / dopaminergic synapse / negative regulation of calcium ion-dependent exocytosis / mu-type opioid receptor binding /  thermoception / Surfactant metabolism / thermoception / Surfactant metabolism /  corticotropin-releasing hormone receptor 1 binding / positive regulation of potassium ion transport / fear response / positive regulation of membrane protein ectodomain proteolysis / corticotropin-releasing hormone receptor 1 binding / positive regulation of potassium ion transport / fear response / positive regulation of membrane protein ectodomain proteolysis /  thioesterase binding / negative regulation of insulin secretion involved in cellular response to glucose stimulus / thioesterase binding / negative regulation of insulin secretion involved in cellular response to glucose stimulus /  norepinephrine binding / norepinephrine binding /  Adrenoceptors / intestinal absorption / positive regulation of epidermal growth factor receptor signaling pathway / dopamine receptor signaling pathway / positive regulation of wound healing / adrenergic receptor signaling pathway / activation of protein kinase activity / Rho protein signal transduction / negative regulation of calcium ion transport / Adrenoceptors / intestinal absorption / positive regulation of epidermal growth factor receptor signaling pathway / dopamine receptor signaling pathway / positive regulation of wound healing / adrenergic receptor signaling pathway / activation of protein kinase activity / Rho protein signal transduction / negative regulation of calcium ion transport /  regulation of vasoconstriction / GABA-ergic synapse / negative regulation of insulin secretion / G protein-coupled serotonin receptor binding / axon terminus / negative regulation of lipid catabolic process / adenylate cyclase-activating adrenergic receptor signaling pathway / presynaptic active zone membrane / cellular response to hormone stimulus / adenylate cyclase-inhibiting G protein-coupled receptor signaling pathway / presynaptic modulation of chemical synaptic transmission / activation of protein kinase B activity / regulation of vasoconstriction / GABA-ergic synapse / negative regulation of insulin secretion / G protein-coupled serotonin receptor binding / axon terminus / negative regulation of lipid catabolic process / adenylate cyclase-activating adrenergic receptor signaling pathway / presynaptic active zone membrane / cellular response to hormone stimulus / adenylate cyclase-inhibiting G protein-coupled receptor signaling pathway / presynaptic modulation of chemical synaptic transmission / activation of protein kinase B activity /  muscle contraction / female pregnancy / positive regulation of cytokine production / postsynaptic density membrane / G-protein beta/gamma-subunit complex binding / Olfactory Signaling Pathway / Activation of the phototransduction cascade / G beta:gamma signalling through PLC beta / Presynaptic function of Kainate receptors / Thromboxane signalling through TP receptor / positive regulation of MAP kinase activity / adenylate cyclase-modulating G protein-coupled receptor signaling pathway / G-protein activation / G protein-coupled acetylcholine receptor signaling pathway / Activation of G protein gated Potassium channels / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / Prostacyclin signalling through prostacyclin receptor / Glucagon signaling in metabolic regulation / G beta:gamma signalling through CDC42 / adenylate cyclase-activating G protein-coupled receptor signaling pathway / ADP signalling through P2Y purinoceptor 12 / G beta:gamma signalling through BTK / Sensory perception of sweet, bitter, and umami (glutamate) taste / Synthesis, secretion, and inactivation of Glucagon-like Peptide-1 (GLP-1) / photoreceptor disc membrane / Adrenaline,noradrenaline inhibits insulin secretion / Glucagon-type ligand receptors / muscle contraction / female pregnancy / positive regulation of cytokine production / postsynaptic density membrane / G-protein beta/gamma-subunit complex binding / Olfactory Signaling Pathway / Activation of the phototransduction cascade / G beta:gamma signalling through PLC beta / Presynaptic function of Kainate receptors / Thromboxane signalling through TP receptor / positive regulation of MAP kinase activity / adenylate cyclase-modulating G protein-coupled receptor signaling pathway / G-protein activation / G protein-coupled acetylcholine receptor signaling pathway / Activation of G protein gated Potassium channels / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / Prostacyclin signalling through prostacyclin receptor / Glucagon signaling in metabolic regulation / G beta:gamma signalling through CDC42 / adenylate cyclase-activating G protein-coupled receptor signaling pathway / ADP signalling through P2Y purinoceptor 12 / G beta:gamma signalling through BTK / Sensory perception of sweet, bitter, and umami (glutamate) taste / Synthesis, secretion, and inactivation of Glucagon-like Peptide-1 (GLP-1) / photoreceptor disc membrane / Adrenaline,noradrenaline inhibits insulin secretion / Glucagon-type ligand receptors /  platelet activation / Vasopressin regulates renal water homeostasis via Aquaporins / G alpha (z) signalling events / cellular response to catecholamine stimulus / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / ADORA2B mediated anti-inflammatory cytokines production / platelet activation / Vasopressin regulates renal water homeostasis via Aquaporins / G alpha (z) signalling events / cellular response to catecholamine stimulus / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / ADORA2B mediated anti-inflammatory cytokines production /  vasodilation / adenylate cyclase-activating dopamine receptor signaling pathway / ADP signalling through P2Y purinoceptor 1 / G beta:gamma signalling through PI3Kgamma / cellular response to prostaglandin E stimulus / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / sensory perception of taste / GPER1 signaling / G-protein beta-subunit binding / vasodilation / adenylate cyclase-activating dopamine receptor signaling pathway / ADP signalling through P2Y purinoceptor 1 / G beta:gamma signalling through PI3Kgamma / cellular response to prostaglandin E stimulus / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / sensory perception of taste / GPER1 signaling / G-protein beta-subunit binding /  heterotrimeric G-protein complex / Inactivation, recovery and regulation of the phototransduction cascade / heterotrimeric G-protein complex / Inactivation, recovery and regulation of the phototransduction cascade /  extracellular vesicle / G alpha (12/13) signalling events / signaling receptor complex adaptor activity / Thrombin signalling through proteinase activated receptors (PARs) / extracellular vesicle / G alpha (12/13) signalling events / signaling receptor complex adaptor activity / Thrombin signalling through proteinase activated receptors (PARs) /  glucose homeostasis / retina development in camera-type eye / glucose homeostasis / retina development in camera-type eye /  GTPase binding / Ca2+ pathway / phospholipase C-activating G protein-coupled receptor signaling pathway / G alpha (i) signalling events GTPase binding / Ca2+ pathway / phospholipase C-activating G protein-coupled receptor signaling pathway / G alpha (i) signalling eventsSimilarity search - Function | |||||||||
| Biological species |   Homo sapiens (human) / Homo sapiens (human) /   Mus musculus (house mouse) Mus musculus (house mouse) | |||||||||
| Method |  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 3.47 Å cryo EM / Resolution: 3.47 Å | |||||||||
 Authors Authors | Xu J / Fink EA / Shoichet BK / Du Y | |||||||||
| Funding support | 1 items
| |||||||||
 Citation Citation |  Journal: Science / Year: 2022 Journal: Science / Year: 2022Title: Structure-based discovery of nonopioid analgesics acting through the α-adrenergic receptor. Authors: Elissa A Fink / Jun Xu / Harald Hübner / Joao M Braz / Philipp Seemann / Charlotte Avet / Veronica Craik / Dorothee Weikert / Maximilian F Schmidt / Chase M Webb / Nataliya A Tolmachova / ...Authors: Elissa A Fink / Jun Xu / Harald Hübner / Joao M Braz / Philipp Seemann / Charlotte Avet / Veronica Craik / Dorothee Weikert / Maximilian F Schmidt / Chase M Webb / Nataliya A Tolmachova / Yurii S Moroz / Xi-Ping Huang / Chakrapani Kalyanaraman / Stefan Gahbauer / Geng Chen / Zheng Liu / Matthew P Jacobson / John J Irwin / Michel Bouvier / Yang Du / Brian K Shoichet / Allan I Basbaum / Peter Gmeiner /       Abstract: Because nonopioid analgesics are much sought after, we computationally docked more than 301 million virtual molecules against a validated pain target, the α-adrenergic receptor (αAR), seeking new ...Because nonopioid analgesics are much sought after, we computationally docked more than 301 million virtual molecules against a validated pain target, the α-adrenergic receptor (αAR), seeking new αAR agonists chemotypes that lack the sedation conferred by known αAR drugs, such as dexmedetomidine. We identified 17 ligands with potencies as low as 12 nanomolar, many with partial agonism and preferential G and G signaling. Experimental structures of αAR complexed with two of these agonists confirmed the docking predictions and templated further optimization. Several compounds, including the initial docking hit '9087 [mean effective concentration (EC) of 52 nanomolar] and two analogs, '7075 and PS75 (EC 4.1 and 4.8 nanomolar), exerted on-target analgesic activity in multiple in vivo pain models without sedation. These newly discovered agonists are interesting as therapeutic leads that lack the liabilities of opioids and the sedation of dexmedetomidine. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_32331.map.gz emd_32331.map.gz | 117.5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-32331-v30.xml emd-32331-v30.xml emd-32331.xml emd-32331.xml | 17 KB 17 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_32331.png emd_32331.png | 115.1 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-32331 http://ftp.pdbj.org/pub/emdb/structures/EMD-32331 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-32331 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-32331 | HTTPS FTP |
-Related structure data
| Related structure data |  7w6pMC  7w7eC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_32331.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_32331.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Cryo-EM structure of the alpha2A adrenergic receptor GoA signaling complex bound to a G protein biased agonist | ||||||||||||||||||||
| Voxel size | X=Y=Z: 0.85 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
- Sample components
Sample components
-Entire : Signaling complex of alpha2A adrenergic receptor with GoA
| Entire | Name: Signaling complex of alpha2A adrenergic receptor with GoA |
|---|---|
| Components |
|
-Supramolecule #1: Signaling complex of alpha2A adrenergic receptor with GoA
| Supramolecule | Name: Signaling complex of alpha2A adrenergic receptor with GoA type: complex / Chimera: Yes / ID: 1 / Parent: 0 / Macromolecule list: #1-#5 |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Recombinant expression | Organism:   Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Recombinant expression | Organism:   Spodoptera frugiperda (fall armyworm) Spodoptera frugiperda (fall armyworm) |
-Macromolecule #1: Guanine nucleotide-binding protein G(o) subunit alpha
| Macromolecule | Name: Guanine nucleotide-binding protein G(o) subunit alpha / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 40.1005 KDa |
| Recombinant expression | Organism:   Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MGCTLSAEER AALERSKAIE KNLKEDGISA AKDVKLLLLG AGESGKSTIV KQMKIIHEDG FSGEDVKQYK PVVYSNTIQS LAAIVRAMD TLGIEYGDKE RKADAKMVCD VVSRMEDTEP FSAELLSAMM RLWGDSGIQE CFNRSREYQL NDSAKYYLDS L DRIGAADY ...String: MGCTLSAEER AALERSKAIE KNLKEDGISA AKDVKLLLLG AGESGKSTIV KQMKIIHEDG FSGEDVKQYK PVVYSNTIQS LAAIVRAMD TLGIEYGDKE RKADAKMVCD VVSRMEDTEP FSAELLSAMM RLWGDSGIQE CFNRSREYQL NDSAKYYLDS L DRIGAADY QPTEQDILRT RVKTTGIVET HFTFKNLHFR LFDVGGQRSE RKKWIHCFED VTAIIFCVAL SGYDQVLHED ET TNRMHES LMLFDSICNN KFFIDTSIIL FLNKKDLFGE KIKKSPLTIC FPEYTGPNTY EDAAAYIQAQ FESKNRSPNK EIY CHMTCA TDTNNIQVVF DAVTDIIIAN NLRGCGLY |
-Macromolecule #2: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 38.402867 KDa |
| Recombinant expression | Organism:   Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: HHHHHHGSSG SELDQLRQEA EQLKNQIRDA RKACADATLS QITNNIDPVG RIQMRTRRTL RGHLAKIYAM HWGTDSRLLV SASQDGKLI IWDSYTTNKV HAIPLRSSWV MTCAYAPSGN YVACGGLDNI CSIYNLKTRE GNVRVSRELA GHTGYLSCCR F LDDNQIVT ...String: HHHHHHGSSG SELDQLRQEA EQLKNQIRDA RKACADATLS QITNNIDPVG RIQMRTRRTL RGHLAKIYAM HWGTDSRLLV SASQDGKLI IWDSYTTNKV HAIPLRSSWV MTCAYAPSGN YVACGGLDNI CSIYNLKTRE GNVRVSRELA GHTGYLSCCR F LDDNQIVT SSGDTTCALW DIETGQQTTT FTGHTGDVMS LSLAPDTRLF VSGACDASAK LWDVREGMCR QTFTGHESDI NA ICFFPNG NAFATGSDDA TCRLFDLRAD QELMTYSHDN IICGITSVSF SKSGRLLLAG YDDFNCNVWD ALKADRAGVL AGH DNRVSC LGVTDDGMAV ATGSWDSFLK IWN |
-Macromolecule #3: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 7.861143 KDa |
| Recombinant expression | Organism:   Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MASNNTASIA QARKLVEQLK MEANIDRIKV SKAAADLMAY CEAHAKEDPL LTPVPASENP FREKKFFCAI L |
-Macromolecule #4: scFv
| Macromolecule | Name: scFv / type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Mus musculus (house mouse) Mus musculus (house mouse) |
| Molecular weight | Theoretical: 32.898781 KDa |
| Recombinant expression | Organism:   Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MLLVNQSHQG FNKEHTSKMV SAIVLYVLLA AAAHSAFADV QLVESGGGLV QPGGSRKLSC SASGFAFSSF GMHWVRQAPE KGLEWVAYI SSGSGTIYYA DTVKGRFTIS RDDPKNTLFL QMTSLRSEDT AMYYCVRSIY YYGSSPFDFW GQGTTLTVSS G GGGSGGGG ...String: MLLVNQSHQG FNKEHTSKMV SAIVLYVLLA AAAHSAFADV QLVESGGGLV QPGGSRKLSC SASGFAFSSF GMHWVRQAPE KGLEWVAYI SSGSGTIYYA DTVKGRFTIS RDDPKNTLFL QMTSLRSEDT AMYYCVRSIY YYGSSPFDFW GQGTTLTVSS G GGGSGGGG SGGGGSDIVM TQATSSVPVT PGESVSISCR SSKSLLHSNG NTYLYWFLQR PGQSPQLLIY RMSNLASGVP DR FSGSGSG TAFTLTISRL EAEDVGVYYC MQHLEYPLTF GAGTKLELKG SLEVLFQGPA AAHHHHHHHH |
-Macromolecule #5: Alpha-2A adrenergic receptor
| Macromolecule | Name: Alpha-2A adrenergic receptor / type: protein_or_peptide / ID: 5 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 50.704566 KDa |
| Recombinant expression | Organism:   Spodoptera frugiperda (fall armyworm) Spodoptera frugiperda (fall armyworm) |
| Sequence | String: MFRQEQPLAE GSFAPMGSLQ PDAGNASWNG TEAPGGGARA TPYSLQVTLT LVCLAGLLML LTVFGNVLVI IAVFTSRALK APQNLFLVS LASADILVAT LVIPFSLANE VMGYWYFGKA WCEIYLALDV LFCTSSIVHL CAISLDRYWS ITQAIEYNLK R TPRRIKAI ...String: MFRQEQPLAE GSFAPMGSLQ PDAGNASWNG TEAPGGGARA TPYSLQVTLT LVCLAGLLML LTVFGNVLVI IAVFTSRALK APQNLFLVS LASADILVAT LVIPFSLANE VMGYWYFGKA WCEIYLALDV LFCTSSIVHL CAISLDRYWS ITQAIEYNLK R TPRRIKAI IITVWVISAV ISFPPLISIE KKGGGGGPQP AEPRCEINDQ KWYVISSCIG SFFAPCLIMI LVYVRIYQIA KR RTRVPPS RRGPDAVAAP PGGTERRPNG LGPERSAGPG GAEAEPLPTQ LNGAPGEPAP AGPRDTDALD LEESSSSDHA ERP PGPRRP ERGPRGKGKA RASQVKPGDS LPRRGPGATG IGTPAAGPGE ERVGAAKASR WRGRQNREKR FTFVLAVVIG VFVV CWFPF FFTYTLTAVG CSVPRTLFKF FFWFGYCNSS LNPVIYTIFN HDFRRAFKKI LCRGDRKRIV |
-Macromolecule #6: N-pyridin-4-ylisoquinolin-4-amine
| Macromolecule | Name: N-pyridin-4-ylisoquinolin-4-amine / type: ligand / ID: 6 / Number of copies: 1 / Formula: W96 |
|---|---|
| Molecular weight | Theoretical: 221.257 Da |
| Chemical component information | 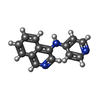 ChemComp-W96: |
-Experimental details
-Structure determination
| Method |  cryo EM cryo EM |
|---|---|
 Processing Processing |  single particle reconstruction single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD Bright-field microscopy / Nominal defocus max: 2.0 µm / Nominal defocus min: 1.0 µm Bright-field microscopy / Nominal defocus max: 2.0 µm / Nominal defocus min: 1.0 µm |
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 60.0 e/Å2 |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: PDB ENTRY PDB model - PDB ID: |
|---|---|
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 3.47 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 287431 |
 Movie
Movie Controller
Controller



























