[English] 日本語
 Yorodumi
Yorodumi- EMDB-30648: Cryo-EM structure of the hE46K cross-seeded mWT alpha-synuclein fibril -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-30648 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of the hE46K cross-seeded mWT alpha-synuclein fibril | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
| Function / homology |  Function and homology information Function and homology informationresponse to desipramine / : / : / : / : / : / response to xenobiotic stimulus => GO:0009410 / : / regulation of neurotransmitter secretion / negative regulation of dopamine metabolic process ...response to desipramine / : / : / : / : / : / response to xenobiotic stimulus => GO:0009410 / : / regulation of neurotransmitter secretion / negative regulation of dopamine metabolic process / membrane organization / neutral lipid metabolic process /  regulation of phospholipase activity / negative regulation of monooxygenase activity / regulation of acyl-CoA biosynthetic process / negative regulation of dopamine uptake involved in synaptic transmission / negative regulation of norepinephrine uptake / positive regulation of glutathione peroxidase activity / positive regulation of SNARE complex assembly / positive regulation of hydrogen peroxide catabolic process / supramolecular fiber / regulation of phospholipase activity / negative regulation of monooxygenase activity / regulation of acyl-CoA biosynthetic process / negative regulation of dopamine uptake involved in synaptic transmission / negative regulation of norepinephrine uptake / positive regulation of glutathione peroxidase activity / positive regulation of SNARE complex assembly / positive regulation of hydrogen peroxide catabolic process / supramolecular fiber /  synaptic transmission, dopaminergic / negative regulation of transporter activity / negative regulation of chaperone-mediated autophagy / mitochondrial membrane organization / cellular response to fibroblast growth factor stimulus / nuclear outer membrane / negative regulation of platelet-derived growth factor receptor signaling pathway / negative regulation of exocytosis / regulation of glutamate secretion / response to iron(II) ion / regulation of norepinephrine uptake / dopamine biosynthetic process / synaptic transmission, dopaminergic / negative regulation of transporter activity / negative regulation of chaperone-mediated autophagy / mitochondrial membrane organization / cellular response to fibroblast growth factor stimulus / nuclear outer membrane / negative regulation of platelet-derived growth factor receptor signaling pathway / negative regulation of exocytosis / regulation of glutamate secretion / response to iron(II) ion / regulation of norepinephrine uptake / dopamine biosynthetic process /  SNARE complex assembly / positive regulation of neurotransmitter secretion / regulation of reactive oxygen species metabolic process / SNARE complex assembly / positive regulation of neurotransmitter secretion / regulation of reactive oxygen species metabolic process /  arachidonic acid binding / synaptic vesicle priming / regulation of locomotion / positive regulation of inositol phosphate biosynthetic process / arachidonic acid binding / synaptic vesicle priming / regulation of locomotion / positive regulation of inositol phosphate biosynthetic process /  regulation of macrophage activation / mitochondrial ATP synthesis coupled electron transport / negative regulation of microtubule polymerization / synaptic vesicle transport / dynein complex binding / positive regulation of receptor recycling / dopamine metabolic process / regulation of dopamine secretion / regulation of macrophage activation / mitochondrial ATP synthesis coupled electron transport / negative regulation of microtubule polymerization / synaptic vesicle transport / dynein complex binding / positive regulation of receptor recycling / dopamine metabolic process / regulation of dopamine secretion /  beta-tubulin binding / negative regulation of thrombin-activated receptor signaling pathway / response to type II interferon / cuprous ion binding / regulation of neuronal synaptic plasticity / positive regulation of exocytosis / synaptic vesicle exocytosis / positive regulation of endocytosis / behavioral response to cocaine / cysteine-type endopeptidase inhibitor activity involved in apoptotic process / beta-tubulin binding / negative regulation of thrombin-activated receptor signaling pathway / response to type II interferon / cuprous ion binding / regulation of neuronal synaptic plasticity / positive regulation of exocytosis / synaptic vesicle exocytosis / positive regulation of endocytosis / behavioral response to cocaine / cysteine-type endopeptidase inhibitor activity involved in apoptotic process /  phospholipase binding / response to magnesium ion / phospholipase binding / response to magnesium ion /  kinesin binding / regulation of presynapse assembly / alpha-tubulin binding / synaptic vesicle endocytosis / negative regulation of serotonin uptake / kinesin binding / regulation of presynapse assembly / alpha-tubulin binding / synaptic vesicle endocytosis / negative regulation of serotonin uptake /  rough endoplasmic reticulum / phospholipid metabolic process / axon terminus / positive regulation of synaptic transmission / rough endoplasmic reticulum / phospholipid metabolic process / axon terminus / positive regulation of synaptic transmission /  inclusion body / cellular response to copper ion / inclusion body / cellular response to copper ion /  Hsp70 protein binding / Hsp70 protein binding /  excitatory postsynaptic potential / response to interleukin-1 / adult locomotory behavior / excitatory postsynaptic potential / response to interleukin-1 / adult locomotory behavior /  SNARE binding / positive regulation of release of sequestered calcium ion into cytosol / fatty acid metabolic process / negative regulation of protein phosphorylation / long-term synaptic potentiation / regulation of transmembrane transporter activity / SNARE binding / positive regulation of release of sequestered calcium ion into cytosol / fatty acid metabolic process / negative regulation of protein phosphorylation / long-term synaptic potentiation / regulation of transmembrane transporter activity /  ferrous iron binding / protein tetramerization / synapse organization / ferrous iron binding / protein tetramerization / synapse organization /  phosphoprotein binding / regulation of long-term neuronal synaptic plasticity / microglial cell activation / phosphoprotein binding / regulation of long-term neuronal synaptic plasticity / microglial cell activation /  phospholipid binding / protein destabilization / negative regulation of cysteine-type endopeptidase activity involved in apoptotic process / cytoplasmic vesicle membrane / tau protein binding / positive regulation of protein serine/threonine kinase activity / phospholipid binding / protein destabilization / negative regulation of cysteine-type endopeptidase activity involved in apoptotic process / cytoplasmic vesicle membrane / tau protein binding / positive regulation of protein serine/threonine kinase activity /  mitochondrial intermembrane space / mitochondrial intermembrane space /  terminal bouton / terminal bouton /  receptor internalization / synaptic vesicle membrane / positive regulation of inflammatory response / activation of cysteine-type endopeptidase activity involved in apoptotic process / receptor internalization / synaptic vesicle membrane / positive regulation of inflammatory response / activation of cysteine-type endopeptidase activity involved in apoptotic process /  synaptic vesicle synaptic vesicleSimilarity search - Function | |||||||||
| Biological species |   Mus musculus (house mouse) Mus musculus (house mouse) | |||||||||
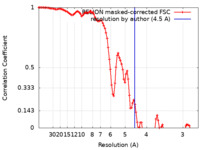
| Method | helical reconstruction /  cryo EM / Resolution: 4.5 Å cryo EM / Resolution: 4.5 Å | |||||||||
 Authors Authors | Long HF / Sun YP / Liu C | |||||||||
 Citation Citation |  Journal: Proc Natl Acad Sci U S A / Year: 2021 Journal: Proc Natl Acad Sci U S A / Year: 2021Title: Wild-type α-synuclein inherits the structure and exacerbated neuropathology of E46K mutant fibril strain by cross-seeding. Authors: Houfang Long / Weitong Zheng / Yang Liu / Yunpeng Sun / Kun Zhao / Zhenying Liu / Wencheng Xia / Shiran Lv / Zhengtao Liu / Dan Li / Kai-Wen He / Cong Liu /  Abstract: Heterozygous point mutations of α-synuclein (α-syn) have been linked to the early onset and rapid progression of familial Parkinson's diseases (fPD). However, the interplay between hereditary ...Heterozygous point mutations of α-synuclein (α-syn) have been linked to the early onset and rapid progression of familial Parkinson's diseases (fPD). However, the interplay between hereditary mutant and wild-type (WT) α-syn and its role in the exacerbated pathology of α-syn in fPD progression are poorly understood. Here, we find that WT mice inoculated with the human E46K mutant α-syn fibril (hE46K) strain develop early-onset motor deficit and morphologically different α-syn aggregation compared with those inoculated with the human WT fibril (hWT) strain. By using cryo-electron microscopy, we reveal at the near-atomic level that the hE46K strain induces both human and mouse WT α-syn monomers to form the fibril structure of the hE46K strain. Moreover, the induced hWT strain inherits most of the pathological traits of the hE46K strain as well. Our work suggests that the structural and pathological features of mutant strains could be propagated by the WT α-syn in such a way that the mutant pathology would be amplified in fPD. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_30648.map.gz emd_30648.map.gz | 6.5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-30648-v30.xml emd-30648-v30.xml emd-30648.xml emd-30648.xml | 8.9 KB 8.9 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_30648_fsc.xml emd_30648_fsc.xml | 10.3 KB | Display |  FSC data file FSC data file |
| Images |  emd_30648.png emd_30648.png | 35 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-30648 http://ftp.pdbj.org/pub/emdb/structures/EMD-30648 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-30648 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-30648 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_30648.map.gz / Format: CCP4 / Size: 91.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_30648.map.gz / Format: CCP4 / Size: 91.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 1.06 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : the hE46K cross-seeded mWT alpha-synuclein fibril
| Entire | Name: the hE46K cross-seeded mWT alpha-synuclein fibril |
|---|---|
| Components |
|
-Supramolecule #1: the hE46K cross-seeded mWT alpha-synuclein fibril
| Supramolecule | Name: the hE46K cross-seeded mWT alpha-synuclein fibril / type: organelle_or_cellular_component / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:   Mus musculus (house mouse) Mus musculus (house mouse) |
| Recombinant expression | Organism:   Escherichia coli (E. coli) Escherichia coli (E. coli) |
-Macromolecule #1: Alpha-synuclein
| Macromolecule | Name: Alpha-synuclein / type: protein_or_peptide / ID: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Mus musculus (house mouse) Mus musculus (house mouse) |
| Recombinant expression | Organism:   Escherichia coli (E. coli) Escherichia coli (E. coli) |
| Sequence | String: MDVFMKGLSK AKEGVVAAAE KTKQGVAEAA GKTKEGVLYV GSKTKEGVVH GVTTVAEKTK EQVTNVGGA VVTGVTAVAQ KTVEGAGNIA AATGFVKKDQ MGKGEEGYPQ EGILEDMPVD P GSEAYEMP SEEGYQDYEP EA |
-Experimental details
-Structure determination
| Method |  cryo EM cryo EM |
|---|---|
 Processing Processing | helical reconstruction |
| Aggregation state | filament |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD Bright-field microscopy Bright-field microscopy |
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Average electron dose: 35.0 e/Å2 |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller