+ Open data
Open data
- Basic information
Basic information
| Entry | 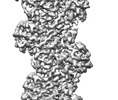 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | T148-ADPR-F-actin | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
| Biological species |   Oryctolagus cuniculus (rabbit) Oryctolagus cuniculus (rabbit) | |||||||||
| Method | helical reconstruction /  cryo EM / Resolution: 3.9 Å cryo EM / Resolution: 3.9 Å | |||||||||
 Authors Authors | Egelman EH | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Int J Mol Sci / Year: 2022 Journal: Int J Mol Sci / Year: 2022Title: TccC3 Toxin Targets the Dynamic Population of F-Actin and Impairs Cell Cortex Integrity. Authors: Songyu Dong / Weili Zheng / Nicholas Pinkerton / Jacob Hansen / Svetlana B Tikunova / Jonathan P Davis / Sarah M Heissler / Elena Kudryashova / Edward H Egelman / Dmitri S Kudryashov /  Abstract: Due to its essential role in cellular processes, actin is a common target for bacterial toxins. One such toxin, TccC3, is an effector domain of the ABC-toxin produced by entomopathogenic bacteria of ...Due to its essential role in cellular processes, actin is a common target for bacterial toxins. One such toxin, TccC3, is an effector domain of the ABC-toxin produced by entomopathogenic bacteria of spp. Unlike other actin-targeting toxins, TccC3 uniquely ADP-ribosylates actin at Thr-148, resulting in the formation of actin aggregates and inhibition of phagocytosis. It has been shown that the fully modified F-actin is resistant to depolymerization by cofilin and gelsolin, but their effects on partially modified actin were not explored. We found that only F-actin unprotected by tropomyosin is the physiological TccC3 substrate. Yet, ADP-ribosylated G-actin can be produced upon cofilin-accelerated F-actin depolymerization, which was only mildly inhibited in partially modified actin. The affinity of TccC3-ADP-ribosylated G-actin for profilin and thymosin-β4 was weakened moderately but sufficiently to potentiate spontaneous polymerization in their presence. Interestingly, the Arp2/3-mediated nucleation was also potentiated by T148-ADP-ribosylation. Notably, even partially modified actin showed reduced bundling by plastins and α-actinin. In agreement with the role of these and other tandem calponin-homology domain actin organizers in the assembly of the cortical actin network, TccC3 induced intense membrane blebbing in cultured cells. Overall, our data suggest that TccC3 imposes a complex action on the cytoskeleton by affecting F-actin nucleation, recycling, and interaction with actin-binding proteins involved in the integration of actin filaments with each other and cellular elements. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_26987.map.gz emd_26987.map.gz | 6.3 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-26987-v30.xml emd-26987-v30.xml emd-26987.xml emd-26987.xml | 14.7 KB 14.7 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_26987.png emd_26987.png | 112.5 KB | ||
| Others |  emd_26987_half_map_1.map.gz emd_26987_half_map_1.map.gz emd_26987_half_map_2.map.gz emd_26987_half_map_2.map.gz | 49.2 MB 49.2 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-26987 http://ftp.pdbj.org/pub/emdb/structures/EMD-26987 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-26987 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-26987 | HTTPS FTP |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_26987.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_26987.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 1.4 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_26987_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_26987_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : F-actin with ADP-ribosylation
| Entire | Name: F-actin with ADP-ribosylation |
|---|---|
| Components |
|
-Supramolecule #1: F-actin with ADP-ribosylation
| Supramolecule | Name: F-actin with ADP-ribosylation / type: organelle_or_cellular_component / ID: 1 / Parent: 0 / Details: Ribosylation due to TccC3 toxin. |
|---|---|
| Source (natural) | Organism:   Oryctolagus cuniculus (rabbit) / Organ: skeletal muscle / Tissue: psoas Oryctolagus cuniculus (rabbit) / Organ: skeletal muscle / Tissue: psoas |
-Experimental details
-Structure determination
| Method |  cryo EM cryo EM |
|---|---|
 Processing Processing | helical reconstruction |
| Aggregation state | filament |
- Sample preparation
Sample preparation
| Concentration | 1.0 mg/mL |
|---|---|
| Buffer | pH: 7 / Component - Concentration: 10.0 mM / Component - Formula: HEPES / Component - Name: F-buffer / Component - Name: F-buffer |
| Grid | Model: EMS Lacey Carbon / Material: COPPER / Mesh: 300 / Support film - Material: CARBON / Support film - topology: HOLEY / Pretreatment - Type: PLASMA CLEANING |
| Vitrification | Cryogen name: ETHANE / Instrument: FEI VITROBOT MARK IV |
| Details | The sample was monodisperse. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated defocus max: 2.5 µm / Calibrated defocus min: 1.5 µm / Calibrated magnification: 55000 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD Bright-field microscopy / Cs: 2.7 mm / Nominal defocus max: 2.5 µm / Nominal defocus min: 1.5 µm / Nominal magnification: 55000 Bright-field microscopy / Cs: 2.7 mm / Nominal defocus max: 2.5 µm / Nominal defocus min: 1.5 µm / Nominal magnification: 55000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Image recording | Film or detector model: FEI FALCON III (4k x 4k) / Digitization - Dimensions - Width: 4096 pixel / Digitization - Dimensions - Height: 4096 pixel / Number grids imaged: 1 / Average electron dose: 50.0 e/Å2 |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Segment selection | Number selected: 250000 / Software - Name: RELION |
|---|---|
| CTF correction | Software - Name: RELION |
| Final angle assignment | Type: NOT APPLICABLE |
| Final reconstruction | Applied symmetry - Helical parameters - Δz: 27.4 Å Applied symmetry - Helical parameters - Δ&Phi: -166.6 ° Applied symmetry - Helical parameters - Axial symmetry: C1 (asymmetric) Resolution.type: BY AUTHOR / Resolution: 3.9 Å / Resolution method: FSC 0.143 CUT-OFF / Software - Name: RELION / Number images used: 250000 |
 Movie
Movie Controller
Controller




 Z
Z Y
Y X
X

















