[English] 日本語
 Yorodumi
Yorodumi- EMDB-19098: Escherichia coli paused disome complex (local refinement of leadi... -
+ Open data
Open data
- Basic information
Basic information
| Entry | 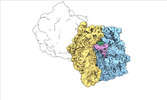 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
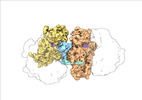
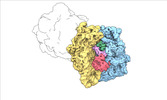
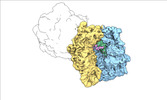
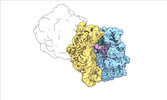
| Title | Escherichia coli paused disome complex (local refinement of leading 70S open non-rotated PRE) | |||||||||
 Map data Map data | Cryo-EM reconstruction of the E. coli disome complex (local refinement of leading 70S open non-rotated PRE). Full map. | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords |  ribosome / ribosome /  polysome / tranlsation / elongation / pausing / disome / polysome / tranlsation / elongation / pausing / disome /  collision / PURE system collision / PURE system | |||||||||
| Biological species |   Escherichia coli (E. coli) Escherichia coli (E. coli) | |||||||||
| Method |  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 3.79 Å cryo EM / Resolution: 3.79 Å | |||||||||
 Authors Authors | Fluegel T / Schacherl M | |||||||||
| Funding support |  Germany, 1 items Germany, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2024 Journal: Nat Commun / Year: 2024Title: Transient disome complex formation in native polysomes during ongoing protein synthesis captured by cryo-EM. Authors: Timo Flügel / Magdalena Schacherl / Anett Unbehaun / Birgit Schroeer / Marylena Dabrowski / Jörg Bürger / Thorsten Mielke / Thiemo Sprink / Christoph A Diebolder / Yollete V Guillén ...Authors: Timo Flügel / Magdalena Schacherl / Anett Unbehaun / Birgit Schroeer / Marylena Dabrowski / Jörg Bürger / Thorsten Mielke / Thiemo Sprink / Christoph A Diebolder / Yollete V Guillén Schlippe / Christian M T Spahn /  Abstract: Structural studies of translating ribosomes traditionally rely on in vitro assembly and stalling of ribosomes in defined states. To comprehensively visualize bacterial translation, we reactivated ex ...Structural studies of translating ribosomes traditionally rely on in vitro assembly and stalling of ribosomes in defined states. To comprehensively visualize bacterial translation, we reactivated ex vivo-derived E. coli polysomes in the PURE in vitro translation system and analyzed the actively elongating polysomes by cryo-EM. We find that 31% of 70S ribosomes assemble into disome complexes that represent eight distinct functional states including decoding and termination intermediates, and a pre-nucleophilic attack state. The functional diversity of disome complexes together with RNase digest experiments suggests that paused disome complexes transiently form during ongoing elongation. Structural analysis revealed five disome interfaces between leading and queueing ribosomes that undergo rearrangements as the leading ribosome traverses through the elongation cycle. Our findings reveal at the molecular level how bL9's CTD obstructs the factor binding site of queueing ribosomes to thwart harmful collisions and illustrate how translation dynamics reshape inter-ribosomal contacts. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_19098.map.gz emd_19098.map.gz | 186.5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-19098-v30.xml emd-19098-v30.xml emd-19098.xml emd-19098.xml | 25.6 KB 25.6 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_19098_fsc.xml emd_19098_fsc.xml | 15.4 KB | Display |  FSC data file FSC data file |
| Images |  emd_19098.png emd_19098.png | 54.8 KB | ||
| Masks |  emd_19098_msk_1.map emd_19098_msk_1.map | 391 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-19098.cif.gz emd-19098.cif.gz | 5.3 KB | ||
| Others |  emd_19098_half_map_1.map.gz emd_19098_half_map_1.map.gz emd_19098_half_map_2.map.gz emd_19098_half_map_2.map.gz | 362.3 MB 362.3 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-19098 http://ftp.pdbj.org/pub/emdb/structures/EMD-19098 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-19098 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-19098 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_19098.map.gz / Format: CCP4 / Size: 391 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_19098.map.gz / Format: CCP4 / Size: 391 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Cryo-EM reconstruction of the E. coli disome complex (local refinement of leading 70S open non-rotated PRE). Full map. | ||||||||||||||||||||
| Voxel size | X=Y=Z: 1.59 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_19098_msk_1.map emd_19098_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Cryo-EM reconstruction of the E. coli disome complex...
| File | emd_19098_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Cryo-EM reconstruction of the E. coli disome complex (local refinement of leading 70S open non-rotated PRE). Half map A. | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Cryo-EM reconstruction of the E. coli disome complex...
| File | emd_19098_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Cryo-EM reconstruction of the E. coli disome complex (local refinement of leading 70S open non-rotated PRE). Half map B. | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Escherichia coli paused disome complex
| Entire | Name: Escherichia coli paused disome complex |
|---|---|
| Components |
|
-Supramolecule #1: Escherichia coli paused disome complex
| Supramolecule | Name: Escherichia coli paused disome complex / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#33, #35-#60 |
|---|---|
| Source (natural) | Organism:   Escherichia coli (E. coli) / Strain: MRE 600 Escherichia coli (E. coli) / Strain: MRE 600 |
-Experimental details
-Structure determination
| Method |  cryo EM cryo EM |
|---|---|
 Processing Processing |  single particle reconstruction single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.6 |
|---|---|
| Grid | Model: Quantifoil R2/2 / Material: COPPER / Mesh: 300 / Support film - Material: CARBON / Support film - topology: HOLEY / Pretreatment - Type: GLOW DISCHARGE |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 75 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV Details: Withdrawn samples were spotted directly onto freshly glow-discharged holey carbon grids, blotted for 1-2 s, and flash frozen in liquid ethane using a Vitrobot Mark IV plunger (ThermoFisher ...Details: Withdrawn samples were spotted directly onto freshly glow-discharged holey carbon grids, blotted for 1-2 s, and flash frozen in liquid ethane using a Vitrobot Mark IV plunger (ThermoFisher Scientific) after a wait time of 40 s at 4 degrees Celcius.. |
| Details | In vitro translation reactions were performed in the PURE translation system using the PURExpress delta ribosome kit (NEB, #E3313S). Translation reactions were supplemented with 0.8 U/uL RNAsin Plus RNase Inhibitor (Promega, N261B). SolA, factor mix, and RNAsin Plus were combined on ice, followed by a preincubation at 37 degrees Celcius for 2 min, and added directly to polysomes (700 nM final concentration) that had been preincubated at 37 degrees Celsius for 2 min. After 1 min reaction time, 4 uL of the reaction mixture were withdrawn for plunge freezing. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 50.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD Bright-field microscopy / Cs: 2.7 mm / Nominal defocus max: 2.0 µm / Nominal defocus min: 0.5 µm / Nominal magnification: 81000 Bright-field microscopy / Cs: 2.7 mm / Nominal defocus max: 2.0 µm / Nominal defocus min: 0.5 µm / Nominal magnification: 81000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Temperature | Min: 80.0 K / Max: 82.0 K |
| Software | Name: EPU (ver. 2.8.1) |
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Number grids imaged: 1 / Number real images: 8982 / Average exposure time: 1.13 sec. / Average electron dose: 45.0 e/Å2 |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
-Atomic model buiding 1
| Initial model | PDB ID: Chain - Source name: PDB / Chain - Initial model type: experimental model |
|---|---|
| Software | Name:  Coot (ver. 0.9.6) Coot (ver. 0.9.6) |
| Details | The atomic model of the E. coli 70S ribosome (PDB ID: 7N1P) was initially docked into the postprocessed density maps with UCSF Chimera and manually adjusted in Coot. All models were refined over multiple rounds using the module phenix.real_space_refine in PHENIX and interactive model building and refinement in Coot, using libG restraints for the RNAs. |
| Refinement | Space: REAL / Protocol: RIGID BODY FIT / Target criteria: Cross-correlation coefficient |
 Movie
Movie Controller
Controller







































 Z
Z Y
Y X
X



























