[English] 日本語
 Yorodumi
Yorodumi- EMDB-16295: Cryo-EM structure of nanodisc-reconstituted wildtype human MRP4 (... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of nanodisc-reconstituted wildtype human MRP4 (in complex with prostaglandin E2) | |||||||||
 Map data Map data | Cryo-EM map for nanodisc-reconstituted wildtype human MRP4 in complex with PGE2 | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | ABC transporter / ABCC4 / MRP4 / PGE2 bound ABC transporter / TRANSLOCASE | |||||||||
| Function / homology |  Function and homology information Function and homology informationguanine nucleotide transmembrane transporter activity / 15-hydroxyprostaglandin dehydrogenase (NAD+) activity / purine nucleotide transmembrane transporter activity / cAMP transport / urate transport / ABC-type bile acid transporter activity / platelet dense granule membrane / leukotriene transport / prostaglandin transport / glutathione transmembrane transporter activity ...guanine nucleotide transmembrane transporter activity / 15-hydroxyprostaglandin dehydrogenase (NAD+) activity / purine nucleotide transmembrane transporter activity / cAMP transport / urate transport / ABC-type bile acid transporter activity / platelet dense granule membrane / leukotriene transport / prostaglandin transport / glutathione transmembrane transporter activity / prostaglandin transmembrane transporter activity / ABC-type glutathione-S-conjugate transporter / ABC-type glutathione S-conjugate transporter activity / urate transmembrane transporter activity / platelet degranulation / ATPase-coupled inorganic anion transmembrane transporter activity / external side of apical plasma membrane / xenobiotic transmembrane transport / export across plasma membrane / ABC-type xenobiotic transporter / prostaglandin secretion / Paracetamol ADME / bile acid and bile salt transport / ABC-type xenobiotic transporter activity / Translocases; Catalysing the translocation of other compounds; Linked to the hydrolysis of a nucleoside triphosphate / Azathioprine ADME / efflux transmembrane transporter activity / xenobiotic transmembrane transporter activity / transport across blood-brain barrier / cilium assembly / ATPase-coupled transmembrane transporter activity / ABC-type transporter activity / xenobiotic metabolic process / ABC-family proteins mediated transport / transmembrane transport / Platelet degranulation / basolateral plasma membrane / apical plasma membrane / nucleolus / Golgi apparatus / ATP hydrolysis activity / ATP binding / membrane / plasma membrane Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 4.0 Å | |||||||||
 Authors Authors | Raj I / Bloch M / Pape TH / Taylor NMI | |||||||||
| Funding support |  Denmark, 1 items Denmark, 1 items
| |||||||||
 Citation Citation |  Journal: Structure / Year: 2023 Journal: Structure / Year: 2023Title: Structural and mechanistic basis of substrate transport by the multidrug transporter MRP4. Authors: Magnus Bloch / Isha Raj / Tillmann Pape / Nicholas M I Taylor /  Abstract: Multidrug resistance-associated protein 4 (MRP4) is an ATP-binding cassette (ABC) transporter expressed at multiple tissue barriers where it actively extrudes a wide variety of drug compounds. ...Multidrug resistance-associated protein 4 (MRP4) is an ATP-binding cassette (ABC) transporter expressed at multiple tissue barriers where it actively extrudes a wide variety of drug compounds. Overexpression of MRP4 provides resistance to clinically used antineoplastic agents, making it a highly attractive therapeutic target for countering multidrug resistance. Here, we report cryo-EM structures of multiple physiologically relevant states of lipid bilayer-embedded human MRP4, including complexes between MRP4 and two widely used chemotherapeutic agents and a complex between MRP4 and its native substrate. The structures display clear similarities and distinct differences in the coordination of these chemically diverse substrates and, in combination with functional and mutational analysis, reveal molecular details of the transport mechanism. Our study provides key insights into the unusually broad substrate specificity of MRP4 and constitutes an important contribution toward a general understanding of multidrug transporters. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_16295.map.gz emd_16295.map.gz | 203.8 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-16295-v30.xml emd-16295-v30.xml emd-16295.xml emd-16295.xml | 18.5 KB 18.5 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_16295_fsc.xml emd_16295_fsc.xml | 12.8 KB | Display |  FSC data file FSC data file |
| Images |  emd_16295.png emd_16295.png | 97.5 KB | ||
| Masks |  emd_16295_msk_1.map emd_16295_msk_1.map | 216 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-16295.cif.gz emd-16295.cif.gz | 6.7 KB | ||
| Others |  emd_16295_half_map_1.map.gz emd_16295_half_map_1.map.gz emd_16295_half_map_2.map.gz emd_16295_half_map_2.map.gz | 200.6 MB 200.6 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-16295 http://ftp.pdbj.org/pub/emdb/structures/EMD-16295 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-16295 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-16295 | HTTPS FTP |
-Validation report
| Summary document |  emd_16295_validation.pdf.gz emd_16295_validation.pdf.gz | 857.9 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_16295_full_validation.pdf.gz emd_16295_full_validation.pdf.gz | 857.4 KB | Display | |
| Data in XML |  emd_16295_validation.xml.gz emd_16295_validation.xml.gz | 21.4 KB | Display | |
| Data in CIF |  emd_16295_validation.cif.gz emd_16295_validation.cif.gz | 27.7 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-16295 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-16295 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-16295 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-16295 | HTTPS FTP |
-Related structure data
| Related structure data |  8bwrMC  8bjfC  8bwoC  8bwpC  8bwqC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_16295.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_16295.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Cryo-EM map for nanodisc-reconstituted wildtype human MRP4 in complex with PGE2 | ||||||||||||||||||||
| Voxel size | X=Y=Z: 0.832 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_16295_msk_1.map emd_16295_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half map 1 for nanodisc-reconstituted wildtype human MRP4...
| File | emd_16295_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map 1 for nanodisc-reconstituted wildtype human MRP4 in complex with PGE2 | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half map 2 for nanodisc-reconstituted wildtype human MRP4...
| File | emd_16295_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map 2 for nanodisc-reconstituted wildtype human MRP4 in complex with PGE2 | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Cryo-EM structure of nanodisc-reconstituted wildtype human MRP4 (...
| Entire | Name: Cryo-EM structure of nanodisc-reconstituted wildtype human MRP4 (in complex with prostaglandin E2) |
|---|---|
| Components |
|
-Supramolecule #1: Cryo-EM structure of nanodisc-reconstituted wildtype human MRP4 (...
| Supramolecule | Name: Cryo-EM structure of nanodisc-reconstituted wildtype human MRP4 (in complex with prostaglandin E2) type: organelle_or_cellular_component / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: ATP-binding cassette sub-family C member 4
| Macromolecule | Name: ATP-binding cassette sub-family C member 4 / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO EC number: Translocases; Catalysing the translocation of other compounds; Linked to the hydrolysis of a nucleoside triphosphate |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 149.693922 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MLPVYQEVKP NPLQDANLCS RVFFWWLNPL FKIGHKRRLE EDDMYSVLPE DRSQHLGEEL QGFWDKEVLR AENDAQKPSL TRAIIKCYW KSYLVLGIFT LIEESAKVIQ PIFLGKIINY FENYDPMDSV ALNTAYAYAT VLTFCTLILA ILHHLYFYHV Q CAGMRLRV ...String: MLPVYQEVKP NPLQDANLCS RVFFWWLNPL FKIGHKRRLE EDDMYSVLPE DRSQHLGEEL QGFWDKEVLR AENDAQKPSL TRAIIKCYW KSYLVLGIFT LIEESAKVIQ PIFLGKIINY FENYDPMDSV ALNTAYAYAT VLTFCTLILA ILHHLYFYHV Q CAGMRLRV AMCHMIYRKA LRLSNMAMGK TTTGQIVNLL SNDVNKFDQV TVFLHFLWAG PLQAIAVTAL LWMEIGISCL AG MAVLIIL LPLQSCFGKL FSSLRSKTAT FTDARIRTMN EVITGIRIIK MYAWEKSFSN LITNLRKKEI SKILRSSCLR GMN LASFFS ASKIIVFVTF TTYVLLGSVI TASRVFVAVT LYGAVRLTVT LFFPSAIERV SEAIVSIRRI QTFLLLDEIS QRNR QLPSD GKKMVHVQDF TAFWDKASET PTLQGLSFTV RPGELLAVVG PVGAGKSSLL SAVLGELAPS HGLVSVHGRI AYVSQ QPWV FSGTLRSNIL FGKKYEKERY EKVIKACALK KDLQLLEDGD LTVIGDRGTT LSGGQKARVN LARAVYQDAD IYLLDD PLS AVDAEVSRHL FELCICQILH EKITILVTHQ LQYLKAASQI LILKDGKMVQ KGTYTEFLKS GIDFGSLLKK DNEESEQ PP VPGTPTLRNR TFSESSVWSQ QSSRPSLKDG ALESQDTENV PVTLSEENRS EGKVGFQAYK NYFRAGAHWI VFIFLILL N TAAQVAYVLQ DWWLSYWANK QSMLNVTVNG GGNVTEKLDL NWYLGIYSGL TVATVLFGIA RSLLVFYVLV NSSQTLHNK MFESILKAPV LFFDRNPIGR ILNRFSKDIG HLDDLLPLTF LDFIQTLLQV VGVVSVAVAV IPWIAIPLVP LGIIFIFLRR YFLETSRDV KRLESTTRSP VFSHLSSSLQ GLWTIRAYKA EERCQELFDA HQDLHSEAWF LFLTTSRWFA VRLDAICAMF V IIVAFGSL ILAKTLDAGQ VGLALSYALT LMGMFQWCVR QSAEVENMMI SVERVIEYTD LEKEAPWEYQ KRPPPAWPHE GV IIFDNVN FMYSPGGPLV LKHLTALIKS QEKVGIVGRT GAGKSSLISA LFRLSEPEGK IWIDKILTTE IGLHDLRKKM SII PQEPVL FTGTMRKNLD PFNEHTDEEL WNALQEVQLK ETIEDLPGKM DTELAESGSN FSVGQRQLVC LARAILRKNQ ILII DEATA NVDPRTDELI QKKIREKFAH CTVLTIAHRL NTIIDSDKIM VLDSGRLKEY DEPYVLLQNK ESLFYKMVQQ LGKAE AAAL TETAKQVYFK RNYPHIGHTD HMVTNTSNGQ PSTLTIFETA L UniProtKB: ATP-binding cassette sub-family C member 4 |
-Macromolecule #2: (Z)-7-[(1R,2R,3R)-3-hydroxy-2-[(E,3S)-3-hydroxyoct-1-enyl]-5-oxo-...
| Macromolecule | Name: (Z)-7-[(1R,2R,3R)-3-hydroxy-2-[(E,3S)-3-hydroxyoct-1-enyl]-5-oxo-cyclopentyl]hept-5-enoic acid type: ligand / ID: 2 / Number of copies: 1 / Formula: P2E |
|---|---|
| Molecular weight | Theoretical: 352.465 Da |
| Chemical component information | 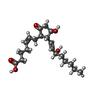 ChemComp-P2E: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Grid | Model: Quantifoil R2/1 / Material: COPPER |
| Vitrification | Cryogen name: ETHANE / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV |
| Details | This sample was monodisperse |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON III (4k x 4k) / Detector mode: COUNTING / Number real images: 3071 / Average electron dose: 40.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.5 µm / Nominal defocus min: 0.5 µm / Nominal magnification: 96000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Protocol: AB INITIO MODEL |
|---|---|
| Output model |  PDB-8bwr: |
 Movie
Movie Controller
Controller










 Z
Z Y
Y X
X


























