[English] 日本語
 Yorodumi
Yorodumi- EMDB-13989: Infectious mouse-adapted RML scrapie prion fibril purified from t... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Infectious mouse-adapted RML scrapie prion fibril purified from terminally-infected mouse brains | |||||||||||||||
 Map data Map data | Ex vivo RML prion fibril | |||||||||||||||
 Sample Sample |
| |||||||||||||||
| Function / homology |  Function and homology information Function and homology informationInsertion of tail-anchored proteins into the endoplasmic reticulum membrane / negative regulation of amyloid precursor protein catabolic process /  lamin binding / regulation of glutamate receptor signaling pathway / regulation of calcium ion import across plasma membrane / aspartic-type endopeptidase inhibitor activity / lamin binding / regulation of glutamate receptor signaling pathway / regulation of calcium ion import across plasma membrane / aspartic-type endopeptidase inhibitor activity /  glycosaminoglycan binding / negative regulation of interleukin-17 production / ATP-dependent protein binding / regulation of potassium ion transmembrane transport ...Insertion of tail-anchored proteins into the endoplasmic reticulum membrane / negative regulation of amyloid precursor protein catabolic process / glycosaminoglycan binding / negative regulation of interleukin-17 production / ATP-dependent protein binding / regulation of potassium ion transmembrane transport ...Insertion of tail-anchored proteins into the endoplasmic reticulum membrane / negative regulation of amyloid precursor protein catabolic process /  lamin binding / regulation of glutamate receptor signaling pathway / regulation of calcium ion import across plasma membrane / aspartic-type endopeptidase inhibitor activity / lamin binding / regulation of glutamate receptor signaling pathway / regulation of calcium ion import across plasma membrane / aspartic-type endopeptidase inhibitor activity /  glycosaminoglycan binding / negative regulation of interleukin-17 production / ATP-dependent protein binding / regulation of potassium ion transmembrane transport / negative regulation of dendritic spine maintenance / type 5 metabotropic glutamate receptor binding / cupric ion binding / nucleobase-containing compound metabolic process / response to copper ion / negative regulation of interleukin-2 production / negative regulation of calcineurin-NFAT signaling cascade / negative regulation of T cell receptor signaling pathway / negative regulation of amyloid-beta formation / cuprous ion binding / activation of protein kinase activity / negative regulation of activated T cell proliferation / negative regulation of long-term synaptic potentiation / response to amyloid-beta / negative regulation of type II interferon production / : / intracellular copper ion homeostasis / positive regulation of protein targeting to membrane / response to cadmium ion / side of membrane / glycosaminoglycan binding / negative regulation of interleukin-17 production / ATP-dependent protein binding / regulation of potassium ion transmembrane transport / negative regulation of dendritic spine maintenance / type 5 metabotropic glutamate receptor binding / cupric ion binding / nucleobase-containing compound metabolic process / response to copper ion / negative regulation of interleukin-2 production / negative regulation of calcineurin-NFAT signaling cascade / negative regulation of T cell receptor signaling pathway / negative regulation of amyloid-beta formation / cuprous ion binding / activation of protein kinase activity / negative regulation of activated T cell proliferation / negative regulation of long-term synaptic potentiation / response to amyloid-beta / negative regulation of type II interferon production / : / intracellular copper ion homeostasis / positive regulation of protein targeting to membrane / response to cadmium ion / side of membrane /  inclusion body / regulation of peptidyl-tyrosine phosphorylation / cellular response to copper ion / neuron projection maintenance / molecular condensate scaffold activity / inclusion body / regulation of peptidyl-tyrosine phosphorylation / cellular response to copper ion / neuron projection maintenance / molecular condensate scaffold activity /  tubulin binding / protein sequestering activity / negative regulation of protein phosphorylation / molecular function activator activity / positive regulation of protein localization to plasma membrane / protein destabilization / protein homooligomerization / negative regulation of DNA-binding transcription factor activity / tubulin binding / protein sequestering activity / negative regulation of protein phosphorylation / molecular function activator activity / positive regulation of protein localization to plasma membrane / protein destabilization / protein homooligomerization / negative regulation of DNA-binding transcription factor activity /  terminal bouton / cellular response to amyloid-beta / positive regulation of neuron apoptotic process / terminal bouton / cellular response to amyloid-beta / positive regulation of neuron apoptotic process /  regulation of protein localization / positive regulation of peptidyl-tyrosine phosphorylation / cellular response to xenobiotic stimulus / regulation of protein localization / positive regulation of peptidyl-tyrosine phosphorylation / cellular response to xenobiotic stimulus /  signaling receptor activity / signaling receptor activity /  amyloid-beta binding / protein-folding chaperone binding / amyloid-beta binding / protein-folding chaperone binding /  microtubule binding / microtubule binding /  nuclear membrane / response to oxidative stress / nuclear membrane / response to oxidative stress /  protease binding / mitochondrial outer membrane / transmembrane transporter binding / protease binding / mitochondrial outer membrane / transmembrane transporter binding /  postsynaptic density / learning or memory / molecular adaptor activity / copper ion binding / postsynaptic density / learning or memory / molecular adaptor activity / copper ion binding /  membrane raft / intracellular membrane-bounded organelle / membrane raft / intracellular membrane-bounded organelle /  dendrite / protein-containing complex binding / negative regulation of apoptotic process / dendrite / protein-containing complex binding / negative regulation of apoptotic process /  Golgi apparatus / Golgi apparatus /  cell surface / cell surface /  endoplasmic reticulum / endoplasmic reticulum /  membrane / identical protein binding / membrane / identical protein binding /  metal ion binding / metal ion binding /  plasma membrane / plasma membrane /  cytosol cytosolSimilarity search - Function | |||||||||||||||
| Biological species |   Mus musculus (house mouse) / Mus musculus (house mouse) /   house mouse (house mouse) house mouse (house mouse) | |||||||||||||||
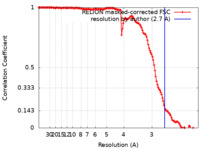
| Method | helical reconstruction /  cryo EM / Resolution: 2.7 Å cryo EM / Resolution: 2.7 Å | |||||||||||||||
 Authors Authors | Manka SW / Zhang W / Wenborn A / Betts J / Joiner S / Saibil HR / Collinge J / Wadsworth JDF | |||||||||||||||
| Funding support |  United Kingdom, 4 items United Kingdom, 4 items
| |||||||||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2022 Journal: Nat Commun / Year: 2022Title: 2.7 Å cryo-EM structure of ex vivo RML prion fibrils. Authors: Szymon W Manka / Wenjuan Zhang / Adam Wenborn / Jemma Betts / Susan Joiner / Helen R Saibil / John Collinge / Jonathan D F Wadsworth /  Abstract: Mammalian prions propagate as distinct strains and are composed of multichain assemblies of misfolded host-encoded prion protein (PrP). Here, we present a near-atomic resolution cryo-EM structure of ...Mammalian prions propagate as distinct strains and are composed of multichain assemblies of misfolded host-encoded prion protein (PrP). Here, we present a near-atomic resolution cryo-EM structure of PrP fibrils present in highly infectious prion rod preparations isolated from the brains of RML prion-infected mice. We found that prion rods comprise single-protofilament helical amyloid fibrils that coexist with twisted pairs of the same protofilaments. Each rung of the protofilament is formed by a single PrP monomer with the ordered core comprising PrP residues 94-225, which folds to create two asymmetric lobes with the N-linked glycans and the glycosylphosphatidylinositol anchor projecting from the C-terminal lobe. The overall architecture is comparable to that of recently reported PrP fibrils isolated from the brain of hamsters infected with the 263K prion strain. However, there are marked conformational variations that could result from differences in PrP sequence and/or represent distinguishing features of the distinct prion strains. | |||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_13989.map.gz emd_13989.map.gz | 10.7 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-13989-v30.xml emd-13989-v30.xml emd-13989.xml emd-13989.xml | 11 KB 11 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_13989_fsc.xml emd_13989_fsc.xml | 13.5 KB | Display |  FSC data file FSC data file |
| Images |  emd_13989.png emd_13989.png | 89.7 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-13989 http://ftp.pdbj.org/pub/emdb/structures/EMD-13989 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-13989 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-13989 | HTTPS FTP |
-Related structure data
| Related structure data |  7qigMC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
| EM raw data |  EMPIAR-10992 (Title: Infectious mouse-adapted RML scrapie prion fibrils purified from terminally-infected mouse brains EMPIAR-10992 (Title: Infectious mouse-adapted RML scrapie prion fibrils purified from terminally-infected mouse brainsData size: 1.0 TB Data #1: Aligned multi-frame micrographs of RML prion fibrils [micrographs - single frame]) |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_13989.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_13989.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Ex vivo RML prion fibril | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.067 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
- Sample components
Sample components
-Entire : Amyloid fibril of mouse PrP from RML--infected mouse brain
| Entire | Name: Amyloid fibril of mouse PrP from RML--infected mouse brain |
|---|---|
| Components |
|
-Supramolecule #1: Amyloid fibril of mouse PrP from RML--infected mouse brain
| Supramolecule | Name: Amyloid fibril of mouse PrP from RML--infected mouse brain type: complex / Chimera: Yes / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:   Mus musculus (house mouse) / Strain: CD-1 / Organ: Brain Mus musculus (house mouse) / Strain: CD-1 / Organ: Brain |
-Macromolecule #1: Major prion protein
| Macromolecule | Name: Major prion protein / type: protein_or_peptide / ID: 1 / Number of copies: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   house mouse (house mouse) / Organ: brain house mouse (house mouse) / Organ: brain |
| Molecular weight | Theoretical: 15.290103 KDa |
| Sequence | String: THNQWNKPSK PKTNLKHVAG AAAAGAVVGG LGGYMLGSAM SRPMIHFGND WEDRYYRENM YRYPNQVYYR PVDQYSNQNN FVHDCVNIT IKQHTVTTTT KGENFTETDV KMMERVVEQM CVTQYQKESQ AYY |
-Experimental details
-Structure determination
| Method |  cryo EM cryo EM |
|---|---|
 Processing Processing | helical reconstruction |
| Aggregation state | filament |
- Sample preparation
Sample preparation
| Buffer | pH: 6.8 |
|---|---|
| Grid | Model: C-flat-2/2 / Material: COPPER / Mesh: 300 / Pretreatment - Type: GLOW DISCHARGE |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD Bright-field microscopy / Nominal defocus max: 3.0 µm / Nominal defocus min: 1.5 µm Bright-field microscopy / Nominal defocus max: 3.0 µm / Nominal defocus min: 1.5 µm |
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 49.0 e/Å2 |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
-Atomic model buiding 1
| Refinement | Space: REAL / Protocol: AB INITIO MODEL |
|---|---|
| Output model |  PDB-7qig: |
 Movie
Movie Controller
Controller





 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)