+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 | データベース: EMDB / ID: EMD-11600 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | Cubic core of the dihydrolipoamide acyltransferase (E2b) component of the branched-chain alpha-ketoacid dehydrogenase complex (BCKDH) from M. tuberculosis | |||||||||
 マップデータ マップデータ | CryoEM sharpened map obtained from Crysoparc | |||||||||
 試料 試料 |
| |||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報 dihydrolipoyllysine-residue (2-methylpropanoyl)transferase / dihydrolipoyllysine-residue (2-methylpropanoyl)transferase /  dihydrolipoyllysine-residue (2-methylpropanoyl)transferase activity / dihydrolipoyllysine-residue (2-methylpropanoyl)transferase activity /  lipoic acid binding / lipoic acid binding /  acetyltransferase activity / peptidoglycan-based cell wall / acetyltransferase activity / peptidoglycan-based cell wall /  細胞膜 / 細胞膜 /  細胞質 細胞質類似検索 - 分子機能 | |||||||||
| 生物種 |   Mycobacterium tuberculosis H37Rv (結核菌) Mycobacterium tuberculosis H37Rv (結核菌) | |||||||||
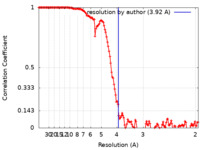
| 手法 |  単粒子再構成法 / 単粒子再構成法 /  クライオ電子顕微鏡法 / 解像度: 3.92 Å クライオ電子顕微鏡法 / 解像度: 3.92 Å | |||||||||
 データ登録者 データ登録者 | Bruch EM / Vilela P / Bellinzoni M | |||||||||
| 資金援助 |  フランス, 1件 フランス, 1件
| |||||||||
 引用 引用 |  ジャーナル: Proc Natl Acad Sci U S A / 年: 2021 ジャーナル: Proc Natl Acad Sci U S A / 年: 2021タイトル: Actinobacteria challenge the paradigm: A unique protein architecture for a well-known, central metabolic complex. 著者: Eduardo M Bruch / Pierre Vilela / Lu Yang / Alexandra Boyko / Norik Lexa-Sapart / Bertrand Raynal / Pedro M Alzari / Marco Bellinzoni /   要旨: α-oxoacid dehydrogenase complexes are large, tripartite enzymatic machineries carrying out key reactions in central metabolism. Extremely conserved across the tree of life, they have been, so far, ...α-oxoacid dehydrogenase complexes are large, tripartite enzymatic machineries carrying out key reactions in central metabolism. Extremely conserved across the tree of life, they have been, so far, all considered to be structured around a high-molecular weight hollow core, consisting of up to 60 subunits of the acyltransferase component. We provide here evidence that Actinobacteria break the rule by possessing an acetyltranferase component reduced to its minimally active, trimeric unit, characterized by a unique C-terminal helix bearing an actinobacterial specific insertion that precludes larger protein oligomerization. This particular feature, together with the presence of an gene coding for both the decarboxylase and the acyltransferase domains on the same polypetide, is spread over Actinobacteria and reflects the association of PDH and ODH into a single physical complex. Considering the central role of the pyruvate and 2-oxoglutarate nodes in central metabolism, our findings pave the way to both therapeutic and metabolic engineering applications. | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| ムービー |
 ムービービューア ムービービューア |
|---|---|
| 構造ビューア | EMマップ:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| 添付画像 |
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_11600.map.gz emd_11600.map.gz | 223.2 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-11600-v30.xml emd-11600-v30.xml emd-11600.xml emd-11600.xml | 14.8 KB 14.8 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| FSC (解像度算出) |  emd_11600_fsc.xml emd_11600_fsc.xml | 13.7 KB | 表示 |  FSCデータファイル FSCデータファイル |
| 画像 |  emd_11600.png emd_11600.png | 203.2 KB | ||
| その他 |  emd_11600_additional_1.map.gz emd_11600_additional_1.map.gz | 10.4 MB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-11600 http://ftp.pdbj.org/pub/emdb/structures/EMD-11600 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-11600 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-11600 | HTTPS FTP |
-関連構造データ
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_11600.map.gz / 形式: CCP4 / 大きさ: 236.9 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_11600.map.gz / 形式: CCP4 / 大きさ: 236.9 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | CryoEM sharpened map obtained from Crysoparc | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 0.97 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 詳細 | EMDB XML:
CCP4マップ ヘッダ情報:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-添付データ
-追加マップ: Map after density modification using ResolveEM in Phenix suite
| ファイル | emd_11600_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Map after density modification using ResolveEM in Phenix suite | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
- 試料の構成要素
試料の構成要素
-全体 : Catalytic domain of M. tuberculosis E2b (acyltransferase from bra...
| 全体 | 名称: Catalytic domain of M. tuberculosis E2b (acyltransferase from branched-chain alpha-ketoacid dehydrogenase complex) |
|---|---|
| 要素 |
|
-超分子 #1: Catalytic domain of M. tuberculosis E2b (acyltransferase from bra...
| 超分子 | 名称: Catalytic domain of M. tuberculosis E2b (acyltransferase from branched-chain alpha-ketoacid dehydrogenase complex) タイプ: complex / ID: 1 / 親要素: 0 / 含まれる分子: all |
|---|---|
| 由来(天然) | 生物種:   Mycobacterium tuberculosis H37Rv (結核菌) Mycobacterium tuberculosis H37Rv (結核菌) |
| 組換発現 | 生物種:   Escherichia coli BL21(DE3) (大腸菌) Escherichia coli BL21(DE3) (大腸菌) |
| 分子量 | 理論値: 592 KDa |
-分子 #1: Dihydrolipoyllysine-residue acyltransferase
| 分子 | 名称: Dihydrolipoyllysine-residue acyltransferase / タイプ: protein_or_peptide / ID: 1 / 光学異性体: LEVO EC番号:  dihydrolipoyllysine-residue (2-methylpropanoyl)transferase dihydrolipoyllysine-residue (2-methylpropanoyl)transferase |
|---|---|
| 由来(天然) | 生物種:   Mycobacterium tuberculosis H37Rv (結核菌) Mycobacterium tuberculosis H37Rv (結核菌) |
| 組換発現 | 生物種:   Escherichia coli BL21(DE3) (大腸菌) Escherichia coli BL21(DE3) (大腸菌) |
| 配列 | 文字列: GSPDVRPVHG VHARMAEK M TLSHKEIPTA KASVEVICAE LLRLRDRFVS AAPEITPFAL TLRLLVIALK HNVILNSTW VDSGEGPQVH VHRGVHLGFG AATERGLLVP VVTDAQDKNT RELASRVAEL ITGAREGTLT PAELRGSTF TVSNFGALGV DDGVPVINHP ...文字列: GSPDVRPVHG VHARMAEK M TLSHKEIPTA KASVEVICAE LLRLRDRFVS AAPEITPFAL TLRLLVIALK HNVILNSTW VDSGEGPQVH VHRGVHLGFG AATERGLLVP VVTDAQDKNT RELASRVAEL ITGAREGTLT PAELRGSTF TVSNFGALGV DDGVPVINHP EAAILGLGAI KPRPVVVGGE VVARPTMTLT C VFDHRVVD GAQVAQFMCE LRDLIESPET ALLDL |
-実験情報
-構造解析
| 手法 |  クライオ電子顕微鏡法 クライオ電子顕微鏡法 |
|---|---|
 解析 解析 |  単粒子再構成法 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 濃度 | 1.6 mg/mL | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 緩衝液 | pH: 8.5 構成要素:
| |||||||||
| グリッド | 材質: COPPER / メッシュ: 200 / 前処理 - タイプ: PLASMA CLEANING / 前処理 - 雰囲気: OTHER / 詳細: Power 5W | |||||||||
| 凍結 | 凍結剤: ETHANE / チャンバー内湿度: 100 % / チャンバー内温度: 277 K / 装置: FEI VITROBOT MARK IV / 詳細: Blot time 4 seconds before plunging. |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | TFS GLACIOS |
|---|---|
| 電子線 | 加速電圧: 200 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | C2レンズ絞り径: 70.0 µm / 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD Bright-field microscopy / Cs: 2.7 mm / 最大 デフォーカス(公称値): 2.4 µm / 最小 デフォーカス(公称値): 0.8 µm / 倍率(公称値): 150000 Bright-field microscopy / Cs: 2.7 mm / 最大 デフォーカス(公称値): 2.4 µm / 最小 デフォーカス(公称値): 0.8 µm / 倍率(公称値): 150000 |
| 試料ステージ | 試料ホルダーモデル: FEI TITAN KRIOS AUTOGRID HOLDER |
| 撮影 | フィルム・検出器のモデル: FEI FALCON III (4k x 4k) 検出モード: INTEGRATING / 撮影したグリッド数: 2 / 実像数: 5009 / 平均露光時間: 1.0 sec. / 平均電子線量: 42.0 e/Å2 |
- 画像解析
画像解析
-原子モデル構築 1
| 初期モデル | PDB ID: Chain - Chain ID: A / Chain - Residue range: 165-393 |
|---|---|
| 精密化 | 空間: REAL / プロトコル: FLEXIBLE FIT |
 ムービー
ムービー コントローラー
コントローラー


















 Z
Z Y
Y X
X