[English] 日本語
 Yorodumi
Yorodumi- EMDB-0322: TnaC-stalled ribosome complex with the titin I27 domain folding c... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-0322 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | TnaC-stalled ribosome complex with the titin I27 domain folding close to the ribosomal exit tunnel | |||||||||
 Map data Map data | Titin I27 domain folded at the exit of ribosomal tunnel | |||||||||
 Sample Sample |
| |||||||||
| Function / homology |  Function and homology information Function and homology informationpositive regulation of tryptophan metabolic process / transcriptional attenuation by ribosome / sarcomerogenesis / structural molecule activity conferring elasticity / telethonin binding / skeletal muscle myosin thick filament assembly / cardiac myofibril assembly / muscle alpha-actinin binding / detection of muscle stretch / tryptophan catabolic process ...positive regulation of tryptophan metabolic process / transcriptional attenuation by ribosome / sarcomerogenesis / structural molecule activity conferring elasticity / telethonin binding / skeletal muscle myosin thick filament assembly / cardiac myofibril assembly / muscle alpha-actinin binding / detection of muscle stretch / tryptophan catabolic process / cardiac muscle tissue morphogenesis / cardiac muscle hypertrophy / mitotic chromosome condensation / Striated Muscle Contraction / actinin binding / M band / I band / protein kinase regulator activity / cardiac muscle cell development / structural constituent of muscle / sarcomere organization / stringent response / skeletal muscle thin filament assembly / striated muscle thin filament / condensed nuclear chromosome / transcriptional attenuation / endoribonuclease inhibitor activity / RNA-binding transcription regulator activity / positive regulation of ribosome biogenesis / negative regulation of cytoplasmic translation / striated muscle contraction / muscle contraction / cardiac muscle contraction / translational termination / DnaA-L2 complex / translation repressor activity / negative regulation of DNA-templated DNA replication initiation / protein kinase A signaling / mRNA regulatory element binding translation repressor activity / ribosome assembly / assembly of large subunit precursor of preribosome / cytosolic ribosome assembly / ribosomal large subunit assembly / response to reactive oxygen species / positive regulation of protein secretion / regulation of cell growth / DNA-templated transcription termination / response to calcium ion / response to radiation / mRNA 5'-UTR binding / Z disc / actin filament binding / large ribosomal subunit / Platelet degranulation / ribosome binding / protein tyrosine kinase activity / transferase activity / 5S rRNA binding / large ribosomal subunit rRNA binding / protease binding / cytosolic large ribosomal subunit / cytoplasmic translation / tRNA binding / calmodulin binding / rRNA binding / non-specific serine/threonine protein kinase / negative regulation of translation / ribosome / structural constituent of ribosome / translation / protein serine kinase activity / response to antibiotic / protein serine/threonine kinase activity / negative regulation of DNA-templated transcription / mRNA binding / calcium ion binding / positive regulation of gene expression / protein kinase binding / enzyme binding / protein homodimerization activity / DNA binding / RNA binding / zinc ion binding / extracellular exosome / extracellular region / ATP binding / identical protein binding / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.2 Å | |||||||||
 Authors Authors | Su T / Kudva R / von Heijne G / Beckmann R | |||||||||
 Citation Citation |  Journal: Proc Natl Acad Sci U S A / Year: 2018 Journal: Proc Natl Acad Sci U S A / Year: 2018Title: Folding pathway of an Ig domain is conserved on and off the ribosome. Authors: Pengfei Tian / Annette Steward / Renuka Kudva / Ting Su / Patrick J Shilling / Adrian A Nickson / Jeffrey J Hollins / Roland Beckmann / Gunnar von Heijne / Jane Clarke / Robert B Best /     Abstract: Proteins that fold cotranslationally may do so in a restricted configurational space, due to the volume occupied by the ribosome. How does this environment, coupled with the close proximity of the ...Proteins that fold cotranslationally may do so in a restricted configurational space, due to the volume occupied by the ribosome. How does this environment, coupled with the close proximity of the ribosome, affect the folding pathway of a protein? Previous studies have shown that the cotranslational folding process for many proteins, including small, single domains, is directly affected by the ribosome. Here, we investigate the cotranslational folding of an all-β Ig domain, titin I27. Using an arrest peptide-based assay and structural studies by cryo-EM, we show that I27 folds in the mouth of the ribosome exit tunnel. Simulations that use a kinetic model for the force dependence of escape from arrest accurately predict the fraction of folded protein as a function of length. We used these simulations to probe the folding pathway on and off the ribosome. Our simulations-which also reproduce experiments on mutant forms of I27-show that I27 folds, while still sequestered in the mouth of the ribosome exit tunnel, by essentially the same pathway as free I27, with only subtle shifts of critical contacts from the C to the N terminus. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_0322.map.gz emd_0322.map.gz | 182.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-0322-v30.xml emd-0322-v30.xml emd-0322.xml emd-0322.xml | 53.3 KB 53.3 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_0322_fsc.xml emd_0322_fsc.xml | 13.2 KB | Display |  FSC data file FSC data file |
| Images |  emd_0322.png emd_0322.png | 181.7 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-0322 http://ftp.pdbj.org/pub/emdb/structures/EMD-0322 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-0322 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-0322 | HTTPS FTP |
-Validation report
| Summary document |  emd_0322_validation.pdf.gz emd_0322_validation.pdf.gz | 289.1 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_0322_full_validation.pdf.gz emd_0322_full_validation.pdf.gz | 288.2 KB | Display | |
| Data in XML |  emd_0322_validation.xml.gz emd_0322_validation.xml.gz | 13.1 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-0322 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-0322 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-0322 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-0322 | HTTPS FTP |
-Related structure data
| Related structure data |  6i0yMC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_0322.map.gz / Format: CCP4 / Size: 196.4 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_0322.map.gz / Format: CCP4 / Size: 196.4 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Titin I27 domain folded at the exit of ribosomal tunnel | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.06 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
+Entire : TnaC-stalled ribosome complex with the titin I27 domain folding c...
+Supramolecule #1: TnaC-stalled ribosome complex with the titin I27 domain folding c...
+Supramolecule #2: ribosome
+Supramolecule #3: Tryptophanase operon leader peptide
+Supramolecule #4: Titin nascent chain
+Macromolecule #1: 50S ribosomal protein L24
+Macromolecule #2: 50S ribosomal protein L32
+Macromolecule #3: 50S ribosomal protein L33
+Macromolecule #4: 50S ribosomal protein L34
+Macromolecule #5: 50S ribosomal protein L35
+Macromolecule #6: 50S ribosomal protein L36
+Macromolecule #7: 50S ribosomal protein L10
+Macromolecule #8: 50S ribosomal protein L7/L12
+Macromolecule #9: Tryptophanase operon leader peptide
+Macromolecule #10: 50S ribosomal protein L25
+Macromolecule #13: 50S ribosomal protein L2
+Macromolecule #14: 50S ribosomal protein L3
+Macromolecule #15: 50S ribosomal protein L4
+Macromolecule #16: 50S ribosomal protein L5
+Macromolecule #17: 50S ribosomal protein L6
+Macromolecule #18: 50S ribosomal protein L9
+Macromolecule #19: 50S ribosomal protein L11
+Macromolecule #20: 50S ribosomal protein L13
+Macromolecule #21: 50S ribosomal protein L14
+Macromolecule #22: 50S ribosomal protein L15
+Macromolecule #23: 50S ribosomal protein L16
+Macromolecule #24: 50S ribosomal protein L17
+Macromolecule #25: 50S ribosomal protein L18
+Macromolecule #26: 50S ribosomal protein L19
+Macromolecule #27: 50S ribosomal protein L20
+Macromolecule #28: 50S ribosomal protein L21
+Macromolecule #29: 50S ribosomal protein L22
+Macromolecule #30: 50S ribosomal protein L23
+Macromolecule #32: 50S ribosomal protein L27
+Macromolecule #33: 50S ribosomal protein L28
+Macromolecule #34: 50S ribosomal protein L29
+Macromolecule #35: 50S ribosomal protein L30
+Macromolecule #36: Titin
+Macromolecule #11: 23S ribosomal RNA
+Macromolecule #12: 5S ribosomal RNA
+Macromolecule #31: Proline tRNA
+Macromolecule #37: MAGNESIUM ION
+Macromolecule #38: ZINC ION
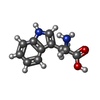
+Macromolecule #39: TRYPTOPHAN
+Macromolecule #40: water
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Grid | Model: Quantifoil R2/2 / Material: COPPER |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277.15 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 QUANTUM (4k x 4k) / Number grids imaged: 1 / Number real images: 2613 / Average electron dose: 0.926 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 70.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 3.0 µm / Nominal defocus min: 1.0 µm |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller