+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-0200 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | human STEAP4 bound to NADPH, FAD and heme. | ||||||||||||
 Map data Map data | None | ||||||||||||
 Sample Sample |
| ||||||||||||
| Function / homology |  Function and homology information Function and homology information ferric-chelate reductase (NADPH) / ferric-chelate reductase (NADPH) /  ferric-chelate reductase (NADPH) activity / cupric reductase activity / iron import into cell / copper ion import / fat cell differentiation / protein homotrimerization / FAD binding / early endosome membrane / ferric-chelate reductase (NADPH) activity / cupric reductase activity / iron import into cell / copper ion import / fat cell differentiation / protein homotrimerization / FAD binding / early endosome membrane /  electron transfer activity ... electron transfer activity ... ferric-chelate reductase (NADPH) / ferric-chelate reductase (NADPH) /  ferric-chelate reductase (NADPH) activity / cupric reductase activity / iron import into cell / copper ion import / fat cell differentiation / protein homotrimerization / FAD binding / early endosome membrane / ferric-chelate reductase (NADPH) activity / cupric reductase activity / iron import into cell / copper ion import / fat cell differentiation / protein homotrimerization / FAD binding / early endosome membrane /  electron transfer activity / electron transfer activity /  endosome / endosome /  Golgi membrane / Golgi membrane /  heme binding / extracellular exosome / heme binding / extracellular exosome /  nucleoplasm / nucleoplasm /  membrane / membrane /  metal ion binding / metal ion binding /  plasma membrane plasma membraneSimilarity search - Function | ||||||||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | ||||||||||||
| Method |  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 3.8 Å cryo EM / Resolution: 3.8 Å | ||||||||||||
 Authors Authors | Oosterheert W / van Bezouwen LS / Rodenburg RNP / Forster F / Mattevi A / Gros P | ||||||||||||
| Funding support |  Netherlands, Netherlands,  Italy, 3 items Italy, 3 items
| ||||||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2018 Journal: Nat Commun / Year: 2018Title: Cryo-EM structures of human STEAP4 reveal mechanism of iron(III) reduction. Authors: Wout Oosterheert / Laura S van Bezouwen / Remco N P Rodenburg / Joke Granneman / Friedrich Förster / Andrea Mattevi / Piet Gros /   Abstract: Enzymes of the six-transmembrane epithelial antigen of the prostate (STEAP) family reduce Fe and Cu ions to facilitate metal-ion uptake by mammalian cells. STEAPs are highly upregulated in several ...Enzymes of the six-transmembrane epithelial antigen of the prostate (STEAP) family reduce Fe and Cu ions to facilitate metal-ion uptake by mammalian cells. STEAPs are highly upregulated in several types of cancer, making them potential therapeutic targets. However, the structural basis for STEAP-catalyzed electron transfer through an array of cofactors to metals at the membrane luminal side remains elusive. Here, we report cryo-electron microscopy structures of human STEAP4 in absence and presence of Fe-NTA. Domain-swapped, trimeric STEAP4 orients NADPH bound to a cytosolic domain onto axially aligned flavin-adenine dinucleotide (FAD) and a single b-type heme that cross the transmembrane-domain to enable electron transfer. Substrate binding within a positively charged ring indicates that iron gets reduced while in complex with its chelator. These molecular principles of iron reduction provide a basis for exploring STEAPs as therapeutic targets. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_0200.map.gz emd_0200.map.gz | 8.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-0200-v30.xml emd-0200-v30.xml emd-0200.xml emd-0200.xml | 24.7 KB 24.7 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_0200_fsc.xml emd_0200_fsc.xml | 8.6 KB | Display |  FSC data file FSC data file |
| Images |  emd_0200.png emd_0200.png | 149.9 KB | ||
| Masks |  emd_0200_msk_1.map emd_0200_msk_1.map | 52.7 MB |  Mask map Mask map | |
| Others |  emd_0200_additional.map.gz emd_0200_additional.map.gz emd_0200_half_map_1.map.gz emd_0200_half_map_1.map.gz emd_0200_half_map_2.map.gz emd_0200_half_map_2.map.gz | 44.5 MB 45.3 MB 45 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-0200 http://ftp.pdbj.org/pub/emdb/structures/EMD-0200 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-0200 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-0200 | HTTPS FTP |
-Related structure data
| Related structure data |  6hd1MC  0199C  6hcyC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_0200.map.gz / Format: CCP4 / Size: 52.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_0200.map.gz / Format: CCP4 / Size: 52.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | None | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.0285 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Mask #1
| File |  emd_0200_msk_1.map emd_0200_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: Unsharpened, unmasked density map of human STEAP4 bound...
| File | emd_0200_additional.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Unsharpened, unmasked density map of human STEAP4 bound to NADPH, FAD and heme (cofactor-bound state). Generated by 3D auto-refine in Relion2.1b1. | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: First half map of human STEAP4 bound to...
| File | emd_0200_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | First half map of human STEAP4 bound to NADPH, FAD and heme (cofactor-bound state). Generated by 3D auto-refine in Relion2.1b1. | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Second half map of human STEAP4 bound to...
| File | emd_0200_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Second half map of human STEAP4 bound to NADPH, FAD and heme (cofactor-bound state). Generated by 3D auto-refine in Relion2.1b1. | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : homotrimer of human STEAP4.
| Entire | Name: homotrimer of human STEAP4. |
|---|---|
| Components |
|
-Supramolecule #1: homotrimer of human STEAP4.
| Supramolecule | Name: homotrimer of human STEAP4. / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Recombinant expression | Organism:   Homo sapiens (human) / Recombinant cell: HEK293 GNTI- / Recombinant plasmid: pUPE 3423 Homo sapiens (human) / Recombinant cell: HEK293 GNTI- / Recombinant plasmid: pUPE 3423 |
| Molecular weight | Theoretical: 152 KDa |
-Macromolecule #1: Metalloreductase STEAP4
| Macromolecule | Name: Metalloreductase STEAP4 / type: protein_or_peptide / ID: 1 / Number of copies: 3 / Enantiomer: LEVO EC number:  Oxidoreductases; Oxidizing metal ions; With NAD+ or NADP+ as acceptor Oxidoreductases; Oxidizing metal ions; With NAD+ or NADP+ as acceptor |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 52.036 KDa |
| Recombinant expression | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MEKTCIDALP LTMNSSEKQE TVCIFGTGDF GRSLGLKMLQ CGYSVVFGSR NPQKTTLLPS GAEVLSYSEA AKKSGIIIIA IHREHYDFL TELTEVLNGK ILVDISNNLK INQYPESNAE YLAHLVPGAH VVKAFNTISA WALQSGALDA SRQVFVCGND S KAKQRVMD ...String: MEKTCIDALP LTMNSSEKQE TVCIFGTGDF GRSLGLKMLQ CGYSVVFGSR NPQKTTLLPS GAEVLSYSEA AKKSGIIIIA IHREHYDFL TELTEVLNGK ILVDISNNLK INQYPESNAE YLAHLVPGAH VVKAFNTISA WALQSGALDA SRQVFVCGND S KAKQRVMD IVRNLGLTPM DQGSLMAAKE IEKYPLQLFP MWRFPFYLSA VLCVFLFFYC VIRDVIYPYV YEKKDNTFRM AI SIPNRIF PITALTLLAL VYLPGVIAAI LQLYRGTKYR RFPDWLDHWM LCRKQLGLVA LGFAFLHVLY TLVIPIRYYV RWR LGNLTV TQAILKKENP FSTSSAWLSD SYVALGILGF FLFVLLGITS LPSVSNAVNW REFRFVQSKL GYLTLILCTA HTLV YGGKR FLSPSNLRWY LPAAYVLGLI IPCTVLVIKF VLIMPCVDNT LTRIRQGWER NSKH |
-Macromolecule #2: NADP NICOTINAMIDE-ADENINE-DINUCLEOTIDE PHOSPHATE
| Macromolecule | Name: NADP NICOTINAMIDE-ADENINE-DINUCLEOTIDE PHOSPHATE / type: ligand / ID: 2 / Number of copies: 3 / Formula: NAP |
|---|---|
| Molecular weight | Theoretical: 743.405 Da |
| Chemical component information |  ChemComp-NAP: |
-Macromolecule #3: PROTOPORPHYRIN IX CONTAINING FE
| Macromolecule | Name: PROTOPORPHYRIN IX CONTAINING FE / type: ligand / ID: 3 / Number of copies: 3 / Formula: HEM |
|---|---|
| Molecular weight | Theoretical: 616.487 Da |
| Chemical component information |  ChemComp-HEM: |
-Macromolecule #4: FLAVIN-ADENINE DINUCLEOTIDE
| Macromolecule | Name: FLAVIN-ADENINE DINUCLEOTIDE / type: ligand / ID: 4 / Number of copies: 3 / Formula: FAD |
|---|---|
| Molecular weight | Theoretical: 785.55 Da |
| Chemical component information |  ChemComp-FAD: |
-Macromolecule #5: 2-acetamido-2-deoxy-beta-D-glucopyranose
| Macromolecule | Name: 2-acetamido-2-deoxy-beta-D-glucopyranose / type: ligand / ID: 5 / Number of copies: 3 / Formula: NAG |
|---|---|
| Molecular weight | Theoretical: 221.208 Da |
| Chemical component information |  ChemComp-NAG: |
-Macromolecule #6: (2R)-3-(phosphonooxy)propane-1,2-diyl dihexanoate
| Macromolecule | Name: (2R)-3-(phosphonooxy)propane-1,2-diyl dihexanoate / type: ligand / ID: 6 / Number of copies: 3 / Formula: 44E |
|---|---|
| Molecular weight | Theoretical: 368.36 Da |
| Chemical component information | 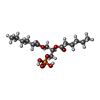 ChemComp-44E: |
-Experimental details
-Structure determination
| Method |  cryo EM cryo EM |
|---|---|
 Processing Processing |  single particle reconstruction single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 4 mg/mL | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 5.5 Component:
Details: 25 mM MES pH 5.5 200 mM NaCl 0.08% digitonin (w/v) | ||||||||||||
| Grid | Model: Quantifoil R1.2/1.3 / Material: GOLD / Mesh: 200 / Pretreatment - Type: GLOW DISCHARGE | ||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 293 K / Instrument: FEI VITROBOT MARK IV / Details: blot time 4seconds blot force 0. | ||||||||||||
| Details | human STEAP4 was purified from the HEK293 GNTI- cell membrane through Strep-affinity chromatography and size-exclusion chromatography (SEC). After SEC in digitonin, the sample was monodisperse. Cofactors NADPH and FAD were added before grid freezing. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TALOS ARCTICA |
|---|---|
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 50.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD Bright-field microscopy / Cs: 2.7 mm / Nominal defocus max: 3.0 µm / Nominal defocus min: 0.8 µm / Nominal magnification: 130000 Bright-field microscopy / Cs: 2.7 mm / Nominal defocus max: 3.0 µm / Nominal defocus min: 0.8 µm / Nominal magnification: 130000 |
| Specialist optics | Energy filter - Slit width: 20 eV |
| Sample stage | Cooling holder cryogen: NITROGEN |
| Details | 200 kV Talos Arctica at Utrecht University, the Netherlands. |
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: SUPER-RESOLUTION / Digitization - Dimensions - Width: 3838 pixel / Digitization - Dimensions - Height: 3710 pixel / Digitization - Frames/image: 1-24 / Number grids imaged: 1 / Number real images: 2321 / Average exposure time: 6.0 sec. / Average electron dose: 45.5 e/Å2 |
| Experimental equipment |  Model: Talos Arctica / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller











 Z
Z Y
Y X
X


































