[English] 日本語
 Yorodumi
Yorodumi- PDB-6hjn: Structure of Influenza Hemagglutinin ectodomain (A/duck/Alberta/35/76) -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6hjn | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of Influenza Hemagglutinin ectodomain (A/duck/Alberta/35/76) | ||||||||||||||||||||||||
 Components Components | (Hemagglutinin ) x 2 ) x 2 | ||||||||||||||||||||||||
 Keywords Keywords |  VIRAL PROTEIN / VIRAL PROTEIN /  Influenza virus / Influenza virus /  Hemagglutinin / Hemagglutinin /  Membrane protein / Membrane protein /  Membrane fusion Membrane fusion | ||||||||||||||||||||||||
| Function / homology |  Function and homology information Function and homology information viral budding from plasma membrane / clathrin-dependent endocytosis of virus by host cell / host cell surface receptor binding / apical plasma membrane / fusion of virus membrane with host plasma membrane / fusion of virus membrane with host endosome membrane / viral budding from plasma membrane / clathrin-dependent endocytosis of virus by host cell / host cell surface receptor binding / apical plasma membrane / fusion of virus membrane with host plasma membrane / fusion of virus membrane with host endosome membrane /  viral envelope / virion attachment to host cell / host cell plasma membrane / virion membrane / viral envelope / virion attachment to host cell / host cell plasma membrane / virion membrane /  membrane membraneSimilarity search - Function | ||||||||||||||||||||||||
| Biological species |    Influenza A virus Influenza A virus | ||||||||||||||||||||||||
| Method |  ELECTRON MICROSCOPY / ELECTRON MICROSCOPY /  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 3.3 Å cryo EM / Resolution: 3.3 Å | ||||||||||||||||||||||||
 Authors Authors | Benton, D.J. / Rosenthal, P.B. | ||||||||||||||||||||||||
| Funding support |  United Kingdom, 7items United Kingdom, 7items
| ||||||||||||||||||||||||
 Citation Citation |  Journal: Proc Natl Acad Sci U S A / Year: 2018 Journal: Proc Natl Acad Sci U S A / Year: 2018Title: Influenza hemagglutinin membrane anchor. Authors: Donald J Benton / Andrea Nans / Lesley J Calder / Jack Turner / Ursula Neu / Yi Pu Lin / Esther Ketelaars / Nicole L Kallewaard / Davide Corti / Antonio Lanzavecchia / Steven J Gamblin / ...Authors: Donald J Benton / Andrea Nans / Lesley J Calder / Jack Turner / Ursula Neu / Yi Pu Lin / Esther Ketelaars / Nicole L Kallewaard / Davide Corti / Antonio Lanzavecchia / Steven J Gamblin / Peter B Rosenthal / John J Skehel /    Abstract: Viruses with membranes fuse them with cellular membranes, to transfer their genomes into cells at the beginning of infection. For Influenza virus, the membrane glycoprotein involved in fusion is the ...Viruses with membranes fuse them with cellular membranes, to transfer their genomes into cells at the beginning of infection. For Influenza virus, the membrane glycoprotein involved in fusion is the hemagglutinin (HA), the 3D structure of which is known from X-ray crystallographic studies. The soluble ectodomain fragments used in these studies lacked the "membrane anchor" portion of the molecule. Since this region has a role in membrane fusion, we have determined its structure by analyzing the intact, full-length molecule in a detergent micelle, using cryo-EM. We have also compared the structures of full-length HA-detergent micelles with full-length HA-Fab complex detergent micelles, to describe an infectivity-neutralizing monoclonal Fab that binds near the ectodomain membrane anchor junction. We determine a high-resolution HA structure which compares favorably in detail with the structure of the ectodomain seen by X-ray crystallography; we detect, clearly, all five carbohydrate side chains of HA; and we find that the ectodomain is joined to the membrane anchor by flexible, eight-residue-long, linkers. The linkers extend into the detergent micelle to join a central triple-helical structure that is a major component of the membrane anchor. | ||||||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6hjn.cif.gz 6hjn.cif.gz | 304 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6hjn.ent.gz pdb6hjn.ent.gz | 253.7 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6hjn.json.gz 6hjn.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/hj/6hjn https://data.pdbj.org/pub/pdb/validation_reports/hj/6hjn ftp://data.pdbj.org/pub/pdb/validation_reports/hj/6hjn ftp://data.pdbj.org/pub/pdb/validation_reports/hj/6hjn | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  0234MC  0235C  0236C  0237C  6hjpC  6hjqC  6hjrC  6hkgC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-Protein , 2 types, 6 molecules ACEBDF
| #1: Protein |  Mass: 35571.695 Da / Num. of mol.: 3 / Source method: isolated from a natural source Source: (natural)   Influenza A virus (strain A/Duck/Alberta/35/1976 H1N1) Influenza A virus (strain A/Duck/Alberta/35/1976 H1N1)References: UniProt: Q9WCE0, UniProt: P26562*PLUS #2: Protein |  Mass: 19838.834 Da / Num. of mol.: 3 / Source method: isolated from a natural source Source: (natural)   Influenza A virus (strain A/Duck/Alberta/35/1976 H1N1) Influenza A virus (strain A/Duck/Alberta/35/1976 H1N1)References: UniProt: P26562 |
|---|
-Sugars , 4 types, 15 molecules
| #3: Polysaccharide | 2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose  / Mass: 424.401 Da / Num. of mol.: 6 / Mass: 424.401 Da / Num. of mol.: 6Source method: isolated from a genetically manipulated source #4: Polysaccharide |  / Mass: 570.542 Da / Num. of mol.: 3 / Mass: 570.542 Da / Num. of mol.: 3Source method: isolated from a genetically manipulated source #5: Polysaccharide |  / Mass: 910.823 Da / Num. of mol.: 3 / Mass: 910.823 Da / Num. of mol.: 3Source method: isolated from a genetically manipulated source #6: Polysaccharide |  / Mass: 586.542 Da / Num. of mol.: 3 / Mass: 586.542 Da / Num. of mol.: 3Source method: isolated from a genetically manipulated source |
|---|
-Non-polymers , 1 types, 3 molecules 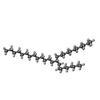
| #7: Chemical |
|---|
-Experimental details
-Experiment
| Experiment | Method:  ELECTRON MICROSCOPY ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method:  single particle reconstruction single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: A/duck/Alberta/35/76 Hemagglutinin Ectodomain / Type: COMPLEX / Entity ID: #1-#2 / Source: NATURAL | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular weight | Value: 0.17 MDa / Experimental value: NO | ||||||||||||||||||||||||||||
| Source (natural) | Organism:   Influenza A virus (strain A/Duck/Alberta/35/1976 H1N1) Influenza A virus (strain A/Duck/Alberta/35/1976 H1N1) | ||||||||||||||||||||||||||||
| Buffer solution | pH: 8 | ||||||||||||||||||||||||||||
| Buffer component |
| ||||||||||||||||||||||||||||
| Specimen | Conc.: 2 mg/ml / Embedding applied: NO / Shadowing applied: NO / Staining applied : NO / Vitrification applied : NO / Vitrification applied : YES : YES | ||||||||||||||||||||||||||||
Vitrification | Instrument: FEI VITROBOT MARK III / Cryogen name: ETHANE |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source : :  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD Bright-field microscopy / Cs Bright-field microscopy / Cs : 2.7 mm / C2 aperture diameter: 50 µm / Alignment procedure: COMA FREE : 2.7 mm / C2 aperture diameter: 50 µm / Alignment procedure: COMA FREE |
| Specimen holder | Cryogen: NITROGEN / Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER |
| Image recording | Electron dose: 43 e/Å2 / Detector mode: COUNTING / Film or detector model: GATAN K2 SUMMIT (4k x 4k) |
| EM imaging optics | Energyfilter name : GIF Quantum LS / Energyfilter slit width: 20 eV : GIF Quantum LS / Energyfilter slit width: 20 eV |
| Image scans | Movie frames/image: 20 |
- Processing
Processing
CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION |
|---|---|
| Symmetry | Point symmetry : C3 (3 fold cyclic : C3 (3 fold cyclic ) ) |
3D reconstruction | Resolution: 3.3 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 365887 / Symmetry type: POINT |
 Movie
Movie Controller
Controller












 PDBj
PDBj






