[English] 日本語
 Yorodumi
Yorodumi- EMDB-30452: Cryo-EM structure of SKF83959 bound dopamine receptor DRD1-Gs sig... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-30452 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of SKF83959 bound dopamine receptor DRD1-Gs signaling complex | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
| Function / homology |  Function and homology information Function and homology informationdopamine neurotransmitter receptor activity / cerebral cortex GABAergic interneuron migration /  Dopamine receptors / dopamine neurotransmitter receptor activity, coupled via Gs / Dopamine receptors / dopamine neurotransmitter receptor activity, coupled via Gs /  operant conditioning / operant conditioning /  dopamine binding / regulation of dopamine uptake involved in synaptic transmission / modification of postsynaptic structure / dopamine binding / regulation of dopamine uptake involved in synaptic transmission / modification of postsynaptic structure /  heterotrimeric G-protein binding / heterotrimeric G-protein binding /  sensitization ...dopamine neurotransmitter receptor activity / cerebral cortex GABAergic interneuron migration / sensitization ...dopamine neurotransmitter receptor activity / cerebral cortex GABAergic interneuron migration /  Dopamine receptors / dopamine neurotransmitter receptor activity, coupled via Gs / Dopamine receptors / dopamine neurotransmitter receptor activity, coupled via Gs /  operant conditioning / operant conditioning /  dopamine binding / regulation of dopamine uptake involved in synaptic transmission / modification of postsynaptic structure / dopamine binding / regulation of dopamine uptake involved in synaptic transmission / modification of postsynaptic structure /  heterotrimeric G-protein binding / heterotrimeric G-protein binding /  sensitization / regulation of dopamine metabolic process / dopamine transport / sensitization / regulation of dopamine metabolic process / dopamine transport /  peristalsis / phospholipase C-activating dopamine receptor signaling pathway / G protein-coupled receptor complex / grooming behavior / positive regulation of neuron migration / peristalsis / phospholipase C-activating dopamine receptor signaling pathway / G protein-coupled receptor complex / grooming behavior / positive regulation of neuron migration /  habituation / astrocyte development / habituation / astrocyte development /  conditioned taste aversion / positive regulation of potassium ion transport / striatum development / dentate gyrus development / conditioned taste aversion / positive regulation of potassium ion transport / striatum development / dentate gyrus development /  maternal behavior / arrestin family protein binding / non-motile cilium / adult walking behavior / mating behavior / long-term synaptic depression / ciliary membrane / dopamine receptor signaling pathway / maternal behavior / arrestin family protein binding / non-motile cilium / adult walking behavior / mating behavior / long-term synaptic depression / ciliary membrane / dopamine receptor signaling pathway /  temperature homeostasis / dopamine metabolic process / transmission of nerve impulse / glucose import / behavioral response to cocaine / PKA activation in glucagon signalling / G protein-coupled receptor signaling pathway, coupled to cyclic nucleotide second messenger / hair follicle placode formation / developmental growth / G-protein alpha-subunit binding / neuronal action potential / temperature homeostasis / dopamine metabolic process / transmission of nerve impulse / glucose import / behavioral response to cocaine / PKA activation in glucagon signalling / G protein-coupled receptor signaling pathway, coupled to cyclic nucleotide second messenger / hair follicle placode formation / developmental growth / G-protein alpha-subunit binding / neuronal action potential /  intracellular transport / GABA-ergic synapse / intracellular transport / GABA-ergic synapse /  D1 dopamine receptor binding / behavioral fear response / positive regulation of cAMP-mediated signaling / Hedgehog 'off' state / D1 dopamine receptor binding / behavioral fear response / positive regulation of cAMP-mediated signaling / Hedgehog 'off' state /  prepulse inhibition / adenylate cyclase-activating adrenergic receptor signaling pathway / prepulse inhibition / adenylate cyclase-activating adrenergic receptor signaling pathway /  synapse assembly / activation of adenylate cyclase activity / response to amphetamine / adenylate cyclase activator activity / presynaptic modulation of chemical synaptic transmission / positive regulation of synaptic transmission, glutamatergic / positive regulation of release of sequestered calcium ion into cytosol / trans-Golgi network membrane / synapse assembly / activation of adenylate cyclase activity / response to amphetamine / adenylate cyclase activator activity / presynaptic modulation of chemical synaptic transmission / positive regulation of synaptic transmission, glutamatergic / positive regulation of release of sequestered calcium ion into cytosol / trans-Golgi network membrane /  synaptic transmission, glutamatergic / G protein-coupled receptor activity / long-term synaptic potentiation / synaptic transmission, glutamatergic / G protein-coupled receptor activity / long-term synaptic potentiation /  regulation of protein phosphorylation / G-protein beta/gamma-subunit complex binding / Olfactory Signaling Pathway / Activation of the phototransduction cascade / regulation of protein phosphorylation / G-protein beta/gamma-subunit complex binding / Olfactory Signaling Pathway / Activation of the phototransduction cascade /  visual learning / visual learning /  bone development / G beta:gamma signalling through PLC beta / Presynaptic function of Kainate receptors / Thromboxane signalling through TP receptor / G-protein activation / G protein-coupled acetylcholine receptor signaling pathway / Activation of G protein gated Potassium channels / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / Prostacyclin signalling through prostacyclin receptor / Glucagon signaling in metabolic regulation / G beta:gamma signalling through CDC42 / adenylate cyclase-activating G protein-coupled receptor signaling pathway / bone development / G beta:gamma signalling through PLC beta / Presynaptic function of Kainate receptors / Thromboxane signalling through TP receptor / G-protein activation / G protein-coupled acetylcholine receptor signaling pathway / Activation of G protein gated Potassium channels / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / Prostacyclin signalling through prostacyclin receptor / Glucagon signaling in metabolic regulation / G beta:gamma signalling through CDC42 / adenylate cyclase-activating G protein-coupled receptor signaling pathway /  cilium / ADP signalling through P2Y purinoceptor 12 / G beta:gamma signalling through BTK / Synthesis, secretion, and inactivation of Glucagon-like Peptide-1 (GLP-1) / cilium / ADP signalling through P2Y purinoceptor 12 / G beta:gamma signalling through BTK / Synthesis, secretion, and inactivation of Glucagon-like Peptide-1 (GLP-1) /  memory / Sensory perception of sweet, bitter, and umami (glutamate) taste / memory / Sensory perception of sweet, bitter, and umami (glutamate) taste /  cognition / photoreceptor disc membrane / cognition / photoreceptor disc membrane /  platelet aggregation / Adrenaline,noradrenaline inhibits insulin secretion / positive regulation of GTPase activity / Glucagon-type ligand receptors / Vasopressin regulates renal water homeostasis via Aquaporins / G alpha (z) signalling events / cellular response to catecholamine stimulus / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / ADORA2B mediated anti-inflammatory cytokines production / sensory perception of taste / ADP signalling through P2Y purinoceptor 1 / adenylate cyclase-activating dopamine receptor signaling pathway / G beta:gamma signalling through PI3Kgamma / cellular response to prostaglandin E stimulus platelet aggregation / Adrenaline,noradrenaline inhibits insulin secretion / positive regulation of GTPase activity / Glucagon-type ligand receptors / Vasopressin regulates renal water homeostasis via Aquaporins / G alpha (z) signalling events / cellular response to catecholamine stimulus / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / ADORA2B mediated anti-inflammatory cytokines production / sensory perception of taste / ADP signalling through P2Y purinoceptor 1 / adenylate cyclase-activating dopamine receptor signaling pathway / G beta:gamma signalling through PI3Kgamma / cellular response to prostaglandin E stimulusSimilarity search - Function | |||||||||
| Biological species |   Homo sapiens (human) / Homo sapiens (human) /   Lama glama (llama) Lama glama (llama) | |||||||||
| Method |  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 3.3 Å cryo EM / Resolution: 3.3 Å | |||||||||
 Authors Authors | Yan W / Shao ZH | |||||||||
 Citation Citation |  Journal: Cell / Year: 2021 Journal: Cell / Year: 2021Title: Ligand recognition and allosteric regulation of DRD1-Gs signaling complexes. Authors: Peng Xiao / Wei Yan / Lu Gou / Ya-Ni Zhong / Liangliang Kong / Chao Wu / Xin Wen / Yuan Yuan / Sheng Cao / Changxiu Qu / Xin Yang / Chuan-Cheng Yang / Anjie Xia / Zhenquan Hu / Qianqian ...Authors: Peng Xiao / Wei Yan / Lu Gou / Ya-Ni Zhong / Liangliang Kong / Chao Wu / Xin Wen / Yuan Yuan / Sheng Cao / Changxiu Qu / Xin Yang / Chuan-Cheng Yang / Anjie Xia / Zhenquan Hu / Qianqian Zhang / Yong-Hao He / Dao-Lai Zhang / Chao Zhang / Gui-Hua Hou / Huanxiang Liu / Lizhe Zhu / Ping Fu / Shengyong Yang / Daniel M Rosenbaum / Jin-Peng Sun / Yang Du / Lei Zhang / Xiao Yu / Zhenhua Shao /   Abstract: Dopamine receptors, including D1- and D2-like receptors, are important therapeutic targets in a variety of neurological syndromes, as well as cardiovascular and kidney diseases. Here, we present five ...Dopamine receptors, including D1- and D2-like receptors, are important therapeutic targets in a variety of neurological syndromes, as well as cardiovascular and kidney diseases. Here, we present five cryoelectron microscopy (cryo-EM) structures of the dopamine D1 receptor (DRD1) coupled to Gs heterotrimer in complex with three catechol-based agonists, a non-catechol agonist, and a positive allosteric modulator for endogenous dopamine. These structures revealed that a polar interaction network is essential for catecholamine-like agonist recognition, whereas specific motifs in the extended binding pocket were responsible for discriminating D1- from D2-like receptors. Moreover, allosteric binding at a distinct inner surface pocket improved the activity of DRD1 by stabilizing endogenous dopamine interaction at the orthosteric site. DRD1-Gs interface revealed key features that serve as determinants for G protein coupling. Together, our study provides a structural understanding of the ligand recognition, allosteric regulation, and G protein coupling mechanisms of DRD1. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_30452.map.gz emd_30452.map.gz | 59.5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-30452-v30.xml emd-30452-v30.xml emd-30452.xml emd-30452.xml | 17.3 KB 17.3 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_30452.png emd_30452.png | 61.8 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-30452 http://ftp.pdbj.org/pub/emdb/structures/EMD-30452 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-30452 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-30452 | HTTPS FTP |
-Related structure data
| Related structure data |  7crhMC  7ckwC  7ckxC  7ckyC  7ckzC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_30452.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_30452.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 0.85 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
+Entire : EM structure of receptor signaling complex-5
+Supramolecule #1: EM structure of receptor signaling complex-5
+Supramolecule #2: Guanine nucleotide-binding protein alpha, beta-1 and gamma-2
+Supramolecule #3: Nanobody35
+Supramolecule #4: D(1A) dopamine receptor
+Macromolecule #1: Guanine nucleotide-binding protein G(s) subunit alpha isoforms short
+Macromolecule #2: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1
+Macromolecule #3: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2
+Macromolecule #4: Nanobody35
+Macromolecule #5: D(1A) dopamine receptor
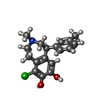
+Macromolecule #6: (1S)-6-chloranyl-3-methyl-1-(3-methylphenyl)-1,2,4,5-tetrahydro-3...
-Experimental details
-Structure determination
| Method |  cryo EM cryo EM |
|---|---|
 Processing Processing |  single particle reconstruction single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: OTHER / Imaging mode: OTHER |
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Average electron dose: 64.0 e/Å2 |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Initial angle assignment | Type: OTHER |
|---|---|
| Final angle assignment | Type: OTHER |
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 3.3 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 140000 |
 Movie
Movie Controller
Controller