+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 |  | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | Tetrahymena Polymerase alpha-Primase | ||||||||||||
 マップデータ マップデータ | full map | ||||||||||||
 試料 試料 |
| ||||||||||||
 キーワード キーワード | CST /  polymerase (ポリメラーゼ) / polymerase (ポリメラーゼ) /  primase (DNAプライマーゼ) / primase (DNAプライマーゼ) /  REPLICATION (DNA複製) REPLICATION (DNA複製) | ||||||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報alpha DNA polymerase:primase complex /  DNA primase activity / lagging strand elongation / DNA primase activity / lagging strand elongation /  DNA replication, synthesis of primer / mitotic DNA replication initiation / leading strand elongation / DNA replication, synthesis of primer / mitotic DNA replication initiation / leading strand elongation /  DNA replication origin binding / DNA replication origin binding /  転移酵素; リンを含む基を移すもの; 核酸を移すもの / 転移酵素; リンを含む基を移すもの; 核酸を移すもの /  single-stranded DNA binding / 4 iron, 4 sulfur cluster binding ...alpha DNA polymerase:primase complex / single-stranded DNA binding / 4 iron, 4 sulfur cluster binding ...alpha DNA polymerase:primase complex /  DNA primase activity / lagging strand elongation / DNA primase activity / lagging strand elongation /  DNA replication, synthesis of primer / mitotic DNA replication initiation / leading strand elongation / DNA replication, synthesis of primer / mitotic DNA replication initiation / leading strand elongation /  DNA replication origin binding / DNA replication origin binding /  転移酵素; リンを含む基を移すもの; 核酸を移すもの / 転移酵素; リンを含む基を移すもの; 核酸を移すもの /  single-stranded DNA binding / 4 iron, 4 sulfur cluster binding / single-stranded DNA binding / 4 iron, 4 sulfur cluster binding /  DNA複製 / DNA複製 /  DNAポリメラーゼ / DNAポリメラーゼ /  DNA-directed DNA polymerase activity / DNA-directed DNA polymerase activity /  nucleotide binding / nucleotide binding /  chromatin binding / chromatin binding /  DNA binding / DNA binding /  metal ion binding / metal ion binding /  細胞核 細胞核類似検索 - 分子機能 | ||||||||||||
| 生物種 |   Tetrahymena thermophila (真核生物) Tetrahymena thermophila (真核生物) | ||||||||||||
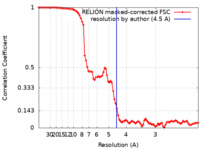
| 手法 |  単粒子再構成法 / 単粒子再構成法 /  クライオ電子顕微鏡法 / 解像度: 4.5 Å クライオ電子顕微鏡法 / 解像度: 4.5 Å | ||||||||||||
 データ登録者 データ登録者 | He Y / Song H / Chan H / Wang Y / Liu B / Susac L / Zhou ZH / Feigon J | ||||||||||||
| 資金援助 |  米国, 3件 米国, 3件
| ||||||||||||
 引用 引用 |  ジャーナル: Nature / 年: 2022 ジャーナル: Nature / 年: 2022タイトル: Structure of Tetrahymena telomerase-bound CST with polymerase α-primase. 著者: Yao He / He Song / Henry Chan / Baocheng Liu / Yaqiang Wang / Lukas Sušac / Z Hong Zhou / Juli Feigon /  要旨: Telomeres are the physical ends of linear chromosomes. They are composed of short repeating sequences (such as TTGGGG in the G-strand for Tetrahymena thermophila) of double-stranded DNA with a single- ...Telomeres are the physical ends of linear chromosomes. They are composed of short repeating sequences (such as TTGGGG in the G-strand for Tetrahymena thermophila) of double-stranded DNA with a single-strand 3' overhang of the G-strand and, in humans, the six shelterin proteins: TPP1, POT1, TRF1, TRF2, RAP1 and TIN2. TPP1 and POT1 associate with the 3' overhang, with POT1 binding the G-strand and TPP1 (in complex with TIN2) recruiting telomerase via interaction with telomerase reverse transcriptase (TERT). The telomere DNA ends are replicated and maintained by telomerase, for the G-strand, and subsequently DNA polymerase α-primase (PolαPrim), for the C-strand. PolαPrim activity is stimulated by the heterotrimeric complex CTC1-STN1-TEN1 (CST), but the structural basis of the recruitment of PolαPrim and CST to telomere ends remains unknown. Here we report cryo-electron microscopy (cryo-EM) structures of Tetrahymena CST in the context of the telomerase holoenzyme, in both the absence and the presence of PolαPrim, and of PolαPrim alone. Tetrahymena Ctc1 binds telomerase subunit p50, a TPP1 orthologue, on a flexible Ctc1 binding motif revealed by cryo-EM and NMR spectroscopy. The PolαPrim polymerase subunit POLA1 binds Ctc1 and Stn1, and its interface with Ctc1 forms an entry port for G-strand DNA to the POLA1 active site. We thus provide a snapshot of four key components that are required for telomeric DNA synthesis in a single active complex-telomerase-core ribonucleoprotein, p50, CST and PolαPrim-that provides insights into the recruitment of CST and PolαPrim and the handoff between G-strand and C-strand synthesis. | ||||||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| 添付画像 |
|---|
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_26867.map.gz emd_26867.map.gz | 49.6 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-26867-v30.xml emd-26867-v30.xml emd-26867.xml emd-26867.xml | 23.9 KB 23.9 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| FSC (解像度算出) |  emd_26867_fsc.xml emd_26867_fsc.xml | 9.1 KB | 表示 |  FSCデータファイル FSCデータファイル |
| 画像 |  emd_26867.png emd_26867.png | 50.7 KB | ||
| マスクデータ |  emd_26867_msk_1.map emd_26867_msk_1.map | 64 MB |  マスクマップ マスクマップ | |
| Filedesc metadata |  emd-26867.cif.gz emd-26867.cif.gz | 7.4 KB | ||
| その他 |  emd_26867_additional_1.map.gz emd_26867_additional_1.map.gz emd_26867_half_map_1.map.gz emd_26867_half_map_1.map.gz emd_26867_half_map_2.map.gz emd_26867_half_map_2.map.gz | 49.1 MB 49.7 MB 49.7 MB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-26867 http://ftp.pdbj.org/pub/emdb/structures/EMD-26867 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-26867 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-26867 | HTTPS FTP |
-関連構造データ
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_26867.map.gz / 形式: CCP4 / 大きさ: 64 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_26867.map.gz / 形式: CCP4 / 大きさ: 64 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | full map | ||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 1.1 Å | ||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||
| 詳細 | EMDB XML:
|
-添付データ
-マスク #1
| ファイル |  emd_26867_msk_1.map emd_26867_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-追加マップ: composite map generated using two focused refined maps, sharpened
| ファイル | emd_26867_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | composite map generated using two focused refined maps, sharpened | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-ハーフマップ: half1
| ファイル | emd_26867_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | half1 | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-ハーフマップ: half2
| ファイル | emd_26867_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | half2 | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
- 試料の構成要素
試料の構成要素
-全体 : Tetrahymena PolaPrim
| 全体 | 名称: Tetrahymena PolaPrim |
|---|---|
| 要素 |
|
-超分子 #1: Tetrahymena PolaPrim
| 超分子 | 名称: Tetrahymena PolaPrim / タイプ: complex / ID: 1 / 親要素: 0 / 含まれる分子: #1-#4 |
|---|---|
| 由来(天然) | 生物種:   Tetrahymena thermophila (真核生物) Tetrahymena thermophila (真核生物) |
-分子 #1: DNA polymerase alpha subunit B
| 分子 | 名称: DNA polymerase alpha subunit B / タイプ: protein_or_peptide / ID: 1 / コピー数: 1 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:   Tetrahymena thermophila (真核生物) Tetrahymena thermophila (真核生物) |
| 分子量 | 理論値: 68.773758 KDa |
| 組換発現 | 生物種:   Spodoptera frugiperda (ツマジロクサヨトウ) Spodoptera frugiperda (ツマジロクサヨトウ) |
| 配列 | 文字列: MEEFEKILEE IDSANLSQNA VQQLKQLIIS NGILDDEQLM EKWLFQLEAI IYNSKQKFLE AQLMDFKKNV LNQDIKKPQN DNFKKIKES EVQKNIDIIK GDADLFTKAS SAKKQTSTQQ ANNAAIDCEE QLRQQLGKES MLDPKRFSYN SNLPQVNFEK N QHQNSNNM ...文字列: MEEFEKILEE IDSANLSQNA VQQLKQLIIS NGILDDEQLM EKWLFQLEAI IYNSKQKFLE AQLMDFKKNV LNQDIKKPQN DNFKKIKES EVQKNIDIIK GDADLFTKAS SAKKQTSTQQ ANNAAIDCEE QLRQQLGKES MLDPKRFSYN SNLPQVNFEK N QHQNSNNM TIVDLFEQYD PNKIPNYLTN NIDKIRKHLE QRILTFKIQN YSQFIVNGEQ LINDVSSYSK TEEVKMVGRL IS TENGFLN TTNLRIELNR NQYFDLVFEN DFDFGNNVLF PNQIVMVKGI INEKEELVVS ELITDHIKEI TDEEHPKIDN LNQ DESQLI MVAAGPFSTV MSTQYTSFDN ILHVAKTKKV STLILLGPFL DIKNEILKDG SIIINSKEYT FEQLQNSLFE KAVK ELEGK TQIIIVPSTR DILSTDSIPQ MSLEIKVSEH KNSIHSYSNP AYIDIDKLRV YIANSDVGMI TLQNSLLDKK IPYMQ QSRL TFKALMNQQN LYSIYPMRNP QDSQQILETV KLPNINDFDI PFDYQLEQYP HIIISPSSLP KFATKIYNTV VINPNY VIR DGSQAGNFAI ITLFKDSDIP IHERTRVDLY CL UniProtKB:  DNA polymerase alpha subunit B DNA polymerase alpha subunit B |
-分子 #2: DNA polymerase
| 分子 | 名称: DNA polymerase / タイプ: protein_or_peptide / ID: 2 / コピー数: 1 / 光学異性体: LEVO / EC番号:  DNAポリメラーゼ DNAポリメラーゼ |
|---|---|
| 由来(天然) | 生物種:   Tetrahymena thermophila (真核生物) Tetrahymena thermophila (真核生物) |
| 分子量 | 理論値: 161.986984 KDa |
| 組換発現 | 生物種:   Spodoptera frugiperda (ツマジロクサヨトウ) Spodoptera frugiperda (ツマジロクサヨトウ) |
| 配列 | 文字列: MSDKLTRLER LNKEVKKQNK LKQHSKNNRF DDDMDIEAYE DDEQIEEDDF IDDTQEDKKY KKKYREIEDE FDQEIEEEEE LNKKKKTKN TILNYTNTTA VTNNKKKAIS KQIPDIDIEE IMKLTERKKK IEQEEAQLLQ EEQELLEQEK REEEEKKRIS Q EAKSILRE ...文字列: MSDKLTRLER LNKEVKKQNK LKQHSKNNRF DDDMDIEAYE DDEQIEEDDF IDDTQEDKKY KKKYREIEDE FDQEIEEEEE LNKKKKTKN TILNYTNTTA VTNNKKKAIS KQIPDIDIEE IMKLTERKKK IEQEEAQLLQ EEQELLEQEK REEEEKKRIS Q EAKSILRE DCASSKTANS SKGKVDQNIL NAINRDFSDD SNTVDSISEF QKLKSLAQKA NLANESLKQS KVSNTEINLT NL SISQVKK INDYKNEDGS VDAYLYDYFY DAQVKPDKIY AFAKVQNKQT NAFDTCVIQI DTIIRNLFFY PSSDTVTEQQ IKN EIAELL KKEQTSRKNV EFLGAFVDKN YAFELPIPRG KSRWYQVVMS YEYEVISPDT KGQYFSYCVG STYSALETFL ITKK ITGPS WVRFQNVKDT TSCITNRKLE FRVDYTNQSN IQVLQKQLPT PPLSVVCISL KTSQQIVLSQ KKKEYKKEIF NLNMK YHEG INIDNSNKDE LNQFKSISFI THIDPTKKQD SITKKGTLPE TTKFCLNELN LLEQFLVHFN EIDPDIVVAH DLYSTV FEI ILTRIREKGI RKWNLLSKLI NIGSSDIPKY GSSTFKTKMA MKGRLLVDTL LSSQEFVNCV EYTLEALAQK LFKIEIP RI DAKAYQQKFA TYKLLNSLVD DTYQDIDYAL RIMYHLQIVP LTKQLTSICG NIWMGSLQNQ RAERNEMLLL HKFNQLNY V YPDNFKNLPE SYKKKHKNAQ IRKQYEEDED QAQGNKNPKK KENKYKGGQV FEPEKGLYNE YIVLLDFNSL YPSIIQEFN VCFTTCVRDP IPLEMQMAPF LGNKKAAIQY SKNQNTKENK MQDEDEEDNE NEQIVQTHDV LPTIEVIKGI APLPSILQYL VEQRKVVKN QIKGQKDPQV IETLDIKQKA FKLVANSMYG CLGFSSSRFY AMPLASFITA KGRHILFDSK KIVEDMGYSV I YGDTDSLM IKPGTNEFLE AVKTGLSIKI KVNSKYKKLQ LDIDGVFKNM LLLKKKKYAT LKVANWEEVK NTNAPEKLEK EI KGIDVVR RDWCQLSRDA GNKILEIILE SKSSENMLDD IKKYLIQLND DINQKNIKNS NYYITKRLTK RVDQYGEKNL PHV AVAQRS IQEKGIDPQT YVNQIISYII CKNEQSSRLV DKAYSPQEFI TQSKSLEIDL QYYKRFQLFE PIKRMLEVIE GINL QEIAS ILEVHYSVQH VSQNNELNAE NVLNLKSKRN QFLTSIPRVL VDCKKCDQTF LFLGILEENA DAASILKCKC GNDIY IQLK NKIALVVKEL IRNFEENAIQ IDNEEFEYTH QISLVGKAKQ QKMSSFTLNQ KLLSIQAMFD ITKEEQENTQ KVTIEK IKT IKKTLDDLLS KSQYNNLNLS NIFTSFGLLK UniProtKB:  DNAポリメラーゼ DNAポリメラーゼ |
-分子 #3: Eukaryotic-type DNA primase, large subunit
| 分子 | 名称: Eukaryotic-type DNA primase, large subunit / タイプ: protein_or_peptide / ID: 3 / コピー数: 1 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:   Tetrahymena thermophila (真核生物) Tetrahymena thermophila (真核生物) |
| 分子量 | 理論値: 64.386645 KDa |
| 組換発現 | 生物種:   Spodoptera frugiperda (ツマジロクサヨトウ) Spodoptera frugiperda (ツマジロクサヨトウ) |
| 配列 | 文字列: MINNKQIQKI QQISKFQLKE YIEMQMQQHS LNKQIKEEER NYILLENAID NSNIQGFQTI QEIVQLAMQR MKMIKEIIQE EPRFNLIDP LSKPNYNRIR VANKFITVAI PKSDPEYELK NKQQYAKEVL SHFLCKMAYA FYAEPETWLE FARAEAFILM D KLKSGQHH ...文字列: MINNKQIQKI QQISKFQLKE YIEMQMQQHS LNKQIKEEER NYILLENAID NSNIQGFQTI QEIVQLAMQR MKMIKEIIQE EPRFNLIDP LSKPNYNRIR VANKFITVAI PKSDPEYELK NKQQYAKEVL SHFLCKMAYA FYAEPETWLE FARAEAFILM D KLKSGQHH ANFFSDENLK IKTISDELFK QAFPKIEATF SSIKIKKNDS EVEQINIKKD NFKMFKFIDH PSMLSNNDVV LH KGYIIIY KESTSKIVQN IFIERLLDEM RLLQLKFQNN GSKLDDDRLS FLKDLHKAEI FNDSTQFNST QIHHYELDRL AKR DMPACM TYLMYGLNQK LHLKHFGRLQ LGLFLKGAGL SLNEALTFWQ KKFSKTSADD FKKKYDYNIR HNYGKLGKQL DYTP MSCQK IIGFQPLKDE FHGCPYKTME SQQLKDFLKL SYNITDEQFV QVNIFKNQKQ YQLACKEVFK VLNDTRDKTK EQFYP YFDK VGNHPNAYFE QSLRMHEPQR FQKQDEEKKQ NKQNRNQNFS ANKQSTNKNN QMDLEF UniProtKB: Eukaryotic-type DNA primase, large subunit |
-分子 #4: DNA primase
| 分子 | 名称: DNA primase / タイプ: protein_or_peptide / ID: 4 / コピー数: 1 / 光学異性体: LEVO EC番号:  転移酵素; リンを含む基を移すもの; 核酸を移すもの 転移酵素; リンを含む基を移すもの; 核酸を移すもの |
|---|---|
| 由来(天然) | 生物種:   Tetrahymena thermophila (真核生物) Tetrahymena thermophila (真核生物) |
| 分子量 | 理論値: 47.172855 KDa |
| 組換発現 | 生物種:   Spodoptera frugiperda (ツマジロクサヨトウ) Spodoptera frugiperda (ツマジロクサヨトウ) |
| 配列 | 文字列: MEIETVEVAP QQEEEFKLDY DLLEEYYRSY FPVSQMVQWL SYPQDGDDTY FTRREFSFTL QNEVYLRYQM YNSEREFKNA LLQKVPEKI DIGAVYDRPG KKGDDIKAKE KEFVIDIDMT DYDHIRTCCS KAKICEKCWK FMRVACDLIS KSLDEDFGFQ H VLWVYSGR ...文字列: MEIETVEVAP QQEEEFKLDY DLLEEYYRSY FPVSQMVQWL SYPQDGDDTY FTRREFSFTL QNEVYLRYQM YNSEREFKNA LLQKVPEKI DIGAVYDRPG KKGDDIKAKE KEFVIDIDMT DYDHIRTCCS KAKICEKCWK FMRVACDLIS KSLDEDFGFQ H VLWVYSGR RGIHAWVCDK EIRKANDYTR ASIIDYLNIL VDNSIGSSYV KPSLLKMEKS HLIERNAMKQ LNQKNDDLKA EK ESEKVFV EIVLREQNLF MKKPEIILEF LAKRSENLSK EVEKEWKTLK TSEQRYEALK ELVSSEDKKK THYLLEELRI WLL YPRLDV NVSKSTNHLL KSPFCIHPKT GNVCVPFTTE EISTFDPFSV PNISTLTTEE GSSKMKNSLK IFNKFLENLK KDV UniProtKB:  DNAプライマーゼ DNAプライマーゼ |
-分子 #5: ZINC ION
| 分子 | 名称: ZINC ION / タイプ: ligand / ID: 5 / コピー数: 1 / 式: ZN |
|---|---|
| 分子量 | 理論値: 65.409 Da |
-実験情報
-構造解析
| 手法 |  クライオ電子顕微鏡法 クライオ電子顕微鏡法 |
|---|---|
 解析 解析 |  単粒子再構成法 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 緩衝液 | pH: 7.5 |
|---|---|
| 凍結 | 凍結剤: ETHANE |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TITAN KRIOS |
|---|---|
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD Bright-field microscopy / 最大 デフォーカス(公称値): 4.0 µm / 最小 デフォーカス(公称値): 0.8 µm Bright-field microscopy / 最大 デフォーカス(公称値): 4.0 µm / 最小 デフォーカス(公称値): 0.8 µm |
| 撮影 | フィルム・検出器のモデル: GATAN K3 (6k x 4k) / 平均電子線量: 55.0 e/Å2 |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
 ムービー
ムービー コントローラー
コントローラー

















 Z
Z Y
Y X
X