+Search query
-Structure paper
| Title | Structure of intact Thermus thermophilus V-ATPase by cryo-EM reveals organization of the membrane-bound V(O) motor. |
|---|---|
| Journal, issue, pages | Proc Natl Acad Sci U S A, Vol. 107, Issue 4, Page 1367-1372, Year 2010 |
| Publish date | Jan 26, 2010 |
 Authors Authors | Wilson C Y Lau / John L Rubinstein /  |
| PubMed Abstract | The eubacterium Thermus thermophilus uses a macromolecular assembly closely related to eukaryotic V-ATPase to produce its supply of ATP. This simplified V-ATPase offers several advantages over ...The eubacterium Thermus thermophilus uses a macromolecular assembly closely related to eukaryotic V-ATPase to produce its supply of ATP. This simplified V-ATPase offers several advantages over eukaryotic V-ATPases for structural analysis and investigation of the mechanism of the enzyme. Here we report the structure of the complex at approximately 16 A resolution as determined by single particle electron cryomicroscopy (cryo-EM). The resolution of the map and our use of cryo-EM, rather than negative stain EM, reveals detailed information about the internal organization of the assembly. We could separate the map into segments corresponding to subunits A and B, the threefold pseudosymmetric C-subunit, a central rotor consisting of subunits D and F, the L-ring, the stator subcomplex consisting of subunits I, E, and G, and a micelle of bound detergent. The architecture of the V(O) region shows a remarkably small area of contact between the I-subunit and the ring of L-subunits and is consistent with a two half-channel model for proton translocation. The arrangement of structural elements in V(O) gives insight into the mechanism of torque generation from proton translocation. |
 External links External links |  Proc Natl Acad Sci U S A / Proc Natl Acad Sci U S A /  PubMed:20080582 / PubMed:20080582 /  PubMed Central PubMed Central |
| Methods | EM (single particle) |
| Resolution | 16.0 Å |
| Structure data | 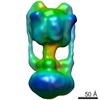 EMDB-1888: |
| Source |
|
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers





 Thermus thermophilus (bacteria)
Thermus thermophilus (bacteria)