+検索条件
-Structure paper
| タイトル | The binding and mechanism of a positive allosteric modulator of Kv3 channels. |
|---|---|
| ジャーナル・号・ページ | Nat Commun, Vol. 15, Issue 1, Page 2533, Year 2024 |
| 掲載日 | 2024年3月21日 |
 著者 著者 | Qiansheng Liang / Gamma Chi / Leonardo Cirqueira / Lianteng Zhi / Agostino Marasco / Nadia Pilati / Martin J Gunthorpe / Giuseppe Alvaro / Charles H Large / David B Sauer / Werner Treptow / Manuel Covarrubias /     |
| PubMed 要旨 | Small-molecule modulators of diverse voltage-gated K (Kv) channels may help treat a wide range of neurological disorders. However, developing effective modulators requires understanding of their ...Small-molecule modulators of diverse voltage-gated K (Kv) channels may help treat a wide range of neurological disorders. However, developing effective modulators requires understanding of their mechanism of action. We apply an orthogonal approach to elucidate the mechanism of action of an imidazolidinedione derivative (AUT5), a highly selective positive allosteric modulator of Kv3.1 and Kv3.2 channels. AUT5 modulation involves positive cooperativity and preferential stabilization of the open state. The cryo-EM structure of the Kv3.1/AUT5 complex at a resolution of 2.5 Å reveals four equivalent AUT5 binding sites at the extracellular inter-subunit interface between the voltage-sensing and pore domains of the channel's tetrameric assembly. Furthermore, we show that the unique extracellular turret regions of Kv3.1 and Kv3.2 essentially govern the selective positive modulation by AUT5. High-resolution apo and bound structures of Kv3.1 demonstrate how AUT5 binding promotes turret rearrangements and interactions with the voltage-sensing domain to favor the open conformation. |
 リンク リンク |  Nat Commun / Nat Commun /  PubMed:38514618 / PubMed:38514618 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) |
| 解像度 | 2.5 - 2.9 Å |
| 構造データ | EMDB-18659, PDB-8quc: EMDB-18660, PDB-8qud: |
| 化合物 | 
ChemComp-WY9: 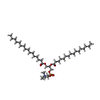 ChemComp-PCF:  ChemComp-ZN:  ChemComp-Y01:  ChemComp-K:  ChemComp-HOH: 
ChemComp-WY0: |
| 由来 |
|
 キーワード キーワード |  MEMBRANE PROTEIN (膜タンパク質) / MEMBRANE PROTEIN (膜タンパク質) /  Modulator (変調方式) / Modulator (変調方式) /  Homotetramer / Homotetramer /  Voltage-gated potassium channel / Kv3.1 / KCNC1 Voltage-gated potassium channel / Kv3.1 / KCNC1 |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について








