+検索条件
-Structure paper
| タイトル | Structural bases of inhibitory mechanism of Ca1.2 channel inhibitors. |
|---|---|
| ジャーナル・号・ページ | Nat Commun, Vol. 15, Issue 1, Page 2772, Year 2024 |
| 掲載日 | 2024年3月30日 |
 著者 著者 | Yiqing Wei / Zhuoya Yu / Lili Wang / Xiaojing Li / Na Li / Qinru Bai / Yuhang Wang / Renjie Li / Yufei Meng / Hao Xu / Xianping Wang / Yanli Dong / Zhuo Huang / Xuejun Cai Zhang / Yan Zhao /  |
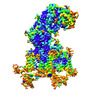
| PubMed 要旨 | The voltage-gated calcium channel Ca1.2 is essential for cardiac and vessel smooth muscle contractility and brain function. Accumulating evidence demonstrates that malfunctions of Ca1.2 are involved ...The voltage-gated calcium channel Ca1.2 is essential for cardiac and vessel smooth muscle contractility and brain function. Accumulating evidence demonstrates that malfunctions of Ca1.2 are involved in brain and heart diseases. Pharmacological inhibition of Ca1.2 is therefore of therapeutic value. Here, we report cryo-EM structures of Ca1.2 in the absence or presence of the antirheumatic drug tetrandrine or antihypertensive drug benidipine. Tetrandrine acts as a pore blocker in a pocket composed of S6, S6, and S6 helices and forms extensive hydrophobic interactions with Ca1.2. Our structure elucidates that benidipine is located in the D-D fenestration site. Its hydrophobic sidechain, phenylpiperidine, is positioned at the exterior of the pore domain and cradled within a hydrophobic pocket formed by S5, S6, and S6 helices, providing additional interactions to exert inhibitory effects on both L-type and T-type voltage gated calcium channels. These findings provide the structural foundation for the rational design and optimization of therapeutic inhibitors of voltage-gated calcium channels. |
 リンク リンク |  Nat Commun / Nat Commun /  PubMed:38555290 / PubMed:38555290 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) |
| 解像度 | 3.3 - 3.5 Å |
| 構造データ | EMDB-34880, PDB-8hlp: EMDB-34891, PDB-8hma: EMDB-34892, PDB-8hmb: |
| 化合物 |  ChemComp-R16:  ChemComp-3PE:  ChemComp-CA:  ChemComp-NAG:  PDB-1ac9:  ChemComp-BMA:  PDB-1ac8: |
| 由来 |
|
 キーワード キーワード |  MEMBRANE PROTEIN (膜タンパク質) / apo state / tet bound state / ben bound state MEMBRANE PROTEIN (膜タンパク質) / apo state / tet bound state / ben bound state |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について