[English] 日本語
 Yorodumi
Yorodumi- EMDB-6432: RCT reconstruction of glycoprotein gHgLgO in complex with neutral... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-6432 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
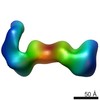
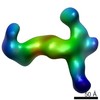
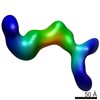
| Title | RCT reconstruction of glycoprotein gHgLgO in complex with neutralizing Fab fragments 3G16 and 13H11 | |||||||||
 Map data Map data | RCT reconstruction of glycoprotein gHgLgO in complex with neutralizing Fab fragments 3G16 and 13H11 | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | CMV / Pentameric gHgLgO entry virus | |||||||||
| Biological species |  Cytomegalovirus / unidentified (others) Cytomegalovirus / unidentified (others) | |||||||||
| Method | single particle reconstruction / Resolution: 19.0 Å | |||||||||
 Authors Authors | Ciferri C / Cianfrocco MA / Chandramouli S / Carfi A | |||||||||
 Citation Citation |  Journal: PLoS Pathog / Year: 2015 Journal: PLoS Pathog / Year: 2015Title: Antigenic Characterization of the HCMV gH/gL/gO and Pentamer Cell Entry Complexes Reveals Binding Sites for Potently Neutralizing Human Antibodies. Authors: Claudio Ciferri / Sumana Chandramouli / Alexander Leitner / Danilo Donnarumma / Michael A Cianfrocco / Rachel Gerrein / Kristian Friedrich / Yukti Aggarwal / Giuseppe Palladino / Ruedi ...Authors: Claudio Ciferri / Sumana Chandramouli / Alexander Leitner / Danilo Donnarumma / Michael A Cianfrocco / Rachel Gerrein / Kristian Friedrich / Yukti Aggarwal / Giuseppe Palladino / Ruedi Aebersold / Nathalie Norais / Ethan C Settembre / Andrea Carfi /    Abstract: Human Cytomegalovirus (HCMV) is a major cause of morbidity and mortality in transplant patients and in fetuses following congenital infection. The glycoprotein complexes gH/gL/gO and ...Human Cytomegalovirus (HCMV) is a major cause of morbidity and mortality in transplant patients and in fetuses following congenital infection. The glycoprotein complexes gH/gL/gO and gH/gL/UL128/UL130/UL131A (Pentamer) are required for HCMV entry in fibroblasts and endothelial/epithelial cells, respectively, and are targeted by potently neutralizing antibodies in the infected host. Using purified soluble forms of gH/gL/gO and Pentamer as well as a panel of naturally elicited human monoclonal antibodies, we determined the location of key neutralizing epitopes on the gH/gL/gO and Pentamer surfaces. Mass Spectrometry (MS) coupled to Chemical Crosslinking or to Hydrogen Deuterium Exchange was used to define residues that are either in proximity or part of neutralizing epitopes on the glycoprotein complexes. We also determined the molecular architecture of the gH/gL/gO- and Pentamer-antibody complexes by Electron Microscopy (EM) and 3D reconstructions. The EM analysis revealed that the Pentamer specific neutralizing antibodies bind to two opposite surfaces of the complex, suggesting that they may neutralize infection by different mechanisms. Together, our data identify the location of neutralizing antibodies binding sites on the gH/gL/gO and Pentamer complexes and provide a framework for the development of antibodies and vaccines against HCMV. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_6432.map.gz emd_6432.map.gz | 3.3 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-6432-v30.xml emd-6432-v30.xml emd-6432.xml emd-6432.xml | 10.8 KB 10.8 KB | Display Display |  EMDB header EMDB header |
| Images |  400_6432.gif 400_6432.gif 80_6432.gif 80_6432.gif | 30.8 KB 3.3 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-6432 http://ftp.pdbj.org/pub/emdb/structures/EMD-6432 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-6432 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-6432 | HTTPS FTP |
-Validation report
| Summary document |  emd_6432_validation.pdf.gz emd_6432_validation.pdf.gz | 78.1 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_6432_full_validation.pdf.gz emd_6432_full_validation.pdf.gz | 77.3 KB | Display | |
| Data in XML |  emd_6432_validation.xml.gz emd_6432_validation.xml.gz | 494 B | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-6432 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-6432 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-6432 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-6432 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_6432.map.gz / Format: CCP4 / Size: 3.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_6432.map.gz / Format: CCP4 / Size: 3.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | RCT reconstruction of glycoprotein gHgLgO in complex with neutralizing Fab fragments 3G16 and 13H11 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 4.3 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Glycoprotein gH/gL/gO Complex bound to 13H11 and 3G16 Fab Fragment
| Entire | Name: Glycoprotein gH/gL/gO Complex bound to 13H11 and 3G16 Fab Fragment |
|---|---|
| Components |
|
-Supramolecule #1000: Glycoprotein gH/gL/gO Complex bound to 13H11 and 3G16 Fab Fragment
| Supramolecule | Name: Glycoprotein gH/gL/gO Complex bound to 13H11 and 3G16 Fab Fragment type: sample / ID: 1000 / Details: Glycosylation increases size. / Number unique components: 5 |
|---|---|
| Molecular weight | Experimental: 300 KDa / Theoretical: 250 KDa / Method: Size Exclusion Chromatography |
-Macromolecule #1: Glycoprotein H
| Macromolecule | Name: Glycoprotein H / type: protein_or_peptide / ID: 1 / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  Cytomegalovirus Cytomegalovirus |
| Molecular weight | Experimental: 300 KDa / Theoretical: 250 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #2: Glycoprotein L
| Macromolecule | Name: Glycoprotein L / type: protein_or_peptide / ID: 2 / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  Cytomegalovirus Cytomegalovirus |
| Molecular weight | Experimental: 300 KDa / Theoretical: 250 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #3: Glycoprotein O
| Macromolecule | Name: Glycoprotein O / type: protein_or_peptide / ID: 3 / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  Cytomegalovirus Cytomegalovirus |
| Molecular weight | Experimental: 300 KDa / Theoretical: 250 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #4: 3G16 antibody Fab fragment
| Macromolecule | Name: 3G16 antibody Fab fragment / type: protein_or_peptide / ID: 4 / Recombinant expression: No / Database: NCBI |
|---|---|
| Source (natural) | Organism: unidentified (others) |
| Molecular weight | Experimental: 300 KDa / Theoretical: 250 KDa |
-Macromolecule #5: 13H11 antibody Fab fragment
| Macromolecule | Name: 13H11 antibody Fab fragment / type: protein_or_peptide / ID: 5 / Recombinant expression: No / Database: NCBI |
|---|---|
| Source (natural) | Organism: unidentified (others) |
| Molecular weight | Experimental: 300 KDa / Theoretical: 250 KDa |
-Experimental details
-Structure determination
 Processing Processing | single particle reconstruction |
|---|---|
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Vitrification | Cryogen name: NONE / Instrument: OTHER |
|---|
- Electron microscopy
Electron microscopy
| Microscope | FEI TECNAI 12 |
|---|---|
| Date | Apr 4, 2014 |
| Image recording | Category: CCD / Film or detector model: FEI EAGLE (4k x 4k) / Number real images: 50 |
| Electron beam | Acceleration voltage: 120 kV / Electron source: LAB6 |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Sample stage | Specimen holder model: SIDE ENTRY, EUCENTRIC |
- Image processing
Image processing
| Final reconstruction | Algorithm: OTHER / Resolution.type: BY AUTHOR / Resolution: 19.0 Å / Resolution method: OTHER / Number images used: 3000 |
|---|
 Movie
Movie Controller
Controller


 UCSF Chimera
UCSF Chimera