+ Open data
Open data
- Basic information
Basic information
| Entry | 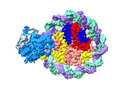 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of DDM1-nucleosome complex in the apo state | |||||||||
 Map data Map data | DDM1-nucleosome complex in the apo state | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords |  complex / complex /  nucleosome / nucleosome /  chromatin remodeling / chromatin remodeling /  structural protein-hydrolase-dna complex / structural protein-hydrolase-dna complex /  GENE REGULATION GENE REGULATION | |||||||||
| Function / homology |  Function and homology information Function and homology informationDNA-mediated transformation /  retrotransposition / chloroplast thylakoid / chromocenter / retrotransposition / chloroplast thylakoid / chromocenter /  thylakoid / response to water deprivation / thylakoid / response to water deprivation /  plasmodesma / plant-type vacuole / DNA methylation-dependent heterochromatin formation / ATP-dependent chromatin remodeler activity ...DNA-mediated transformation / plasmodesma / plant-type vacuole / DNA methylation-dependent heterochromatin formation / ATP-dependent chromatin remodeler activity ...DNA-mediated transformation /  retrotransposition / chloroplast thylakoid / chromocenter / retrotransposition / chloroplast thylakoid / chromocenter /  thylakoid / response to water deprivation / thylakoid / response to water deprivation /  plasmodesma / plant-type vacuole / DNA methylation-dependent heterochromatin formation / ATP-dependent chromatin remodeler activity / plasmodesma / plant-type vacuole / DNA methylation-dependent heterochromatin formation / ATP-dependent chromatin remodeler activity /  plastid / plastid /  chloroplast stroma / heterochromatin formation / epigenetic regulation of gene expression / chloroplast stroma / heterochromatin formation / epigenetic regulation of gene expression /  DNA helicase activity / DNA helicase activity /  chloroplast / response to bacterium / response to wounding / chloroplast / response to bacterium / response to wounding /  peroxisome / structural constituent of chromatin / peroxisome / structural constituent of chromatin /  nucleosome / nucleosome /  DNA helicase / DNA helicase /  chromatin remodeling / protein heterodimerization activity / chromatin remodeling / protein heterodimerization activity /  nucleolus / nucleolus /  ATP hydrolysis activity / ATP hydrolysis activity /  DNA binding / extracellular region / DNA binding / extracellular region /  ATP binding / ATP binding /  nucleus / nucleus /  plasma membrane / plasma membrane /  cytosol cytosolSimilarity search - Function | |||||||||
| Biological species |   Arabidopsis thaliana (thale cress) / synthetic construct (others) Arabidopsis thaliana (thale cress) / synthetic construct (others) | |||||||||
| Method |  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 3.58 Å cryo EM / Resolution: 3.58 Å | |||||||||
 Authors Authors | Liu Y / Zhang Z / Du J | |||||||||
| Funding support |  China, 1 items China, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Plants / Year: 2024 Journal: Nat Plants / Year: 2024Title: Molecular basis of chromatin remodelling by DDM1 involved in plant DNA methylation. Authors: Yue Liu / Zhihui Zhang / Hongmiao Hu / Wei Chen / Fan Zhang / Qian Wang / Changshi Wang / Kaige Yan / Jiamu Du /   Abstract: Eukaryotic gene regulation occurs at the chromatin level, which requires changing the chromatin structure by a group of ATP-dependent DNA translocases-namely, the chromatin remodellers. In plants, ...Eukaryotic gene regulation occurs at the chromatin level, which requires changing the chromatin structure by a group of ATP-dependent DNA translocases-namely, the chromatin remodellers. In plants, chromatin remodellers function in various biological processes and possess both conserved and plant-specific components. DECREASE IN DNA METHYLATION 1 (DDM1) is a plant chromatin remodeller that plays a key role in the maintenance DNA methylation. Here we determined the structures of Arabidopsis DDM1 in complex with nucleosome in ADP-BeF-bound, ADP-bound and nucleotide-free conformations. We show that DDM1 specifically recognizes the H4 tail and nucleosomal DNA. The conformational differences between ADP-BeF-bound, ADP-bound and nucleotide-free DDM1 suggest a chromatin remodelling cycle coupled to ATP binding, hydrolysis and ADP release. This, in turn, triggers conformational changes in the DDM1-bound nucleosomal DNA, which alters the nucleosome structure and promotes DNA sliding. Together, our data reveal the molecular basis of chromatin remodelling by DDM1. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_37529.map.gz emd_37529.map.gz | 7.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-37529-v30.xml emd-37529-v30.xml emd-37529.xml emd-37529.xml | 23 KB 23 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_37529_fsc.xml emd_37529_fsc.xml | 9.2 KB | Display |  FSC data file FSC data file |
| Images |  emd_37529.png emd_37529.png | 97.9 KB | ||
| Filedesc metadata |  emd-37529.cif.gz emd-37529.cif.gz | 7.3 KB | ||
| Others |  emd_37529_half_map_1.map.gz emd_37529_half_map_1.map.gz emd_37529_half_map_2.map.gz emd_37529_half_map_2.map.gz | 49.4 MB 49.6 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-37529 http://ftp.pdbj.org/pub/emdb/structures/EMD-37529 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-37529 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-37529 | HTTPS FTP |
-Related structure data
| Related structure data |  8wh5MC  8wh8C  8wh9C  8whaC  8whbC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_37529.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_37529.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | DDM1-nucleosome complex in the apo state | ||||||||||||||||||||
| Voxel size | X=Y=Z: 0.855 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: Half1 map of DDM1-nucleosome complex in the apo state
| File | emd_37529_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half1 map of DDM1-nucleosome complex in the apo state | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half2 map of DDM1-nucleosome complex in the apo state
| File | emd_37529_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half2 map of DDM1-nucleosome complex in the apo state | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : DDM1-nucleosome complex in the apo state
| Entire | Name: DDM1-nucleosome complex in the apo state |
|---|---|
| Components |
|
-Supramolecule #1: DDM1-nucleosome complex in the apo state
| Supramolecule | Name: DDM1-nucleosome complex in the apo state / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#7 |
|---|---|
| Source (natural) | Organism:   Arabidopsis thaliana (thale cress) Arabidopsis thaliana (thale cress) |
| Molecular weight | Theoretical: 290 KDa |
-Macromolecule #1: Histone H3.1
| Macromolecule | Name: Histone H3.1 / type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Arabidopsis thaliana (thale cress) Arabidopsis thaliana (thale cress) |
| Molecular weight | Theoretical: 15.300968 KDa |
| Recombinant expression | Organism:   Escherichia coli BL21 (bacteria) Escherichia coli BL21 (bacteria) |
| Sequence | String: MARTKQTARK STGGKAPRKQ LATKAARKSA PATGGVKKPH RFRPGTVALR EIRKYQKSTE LLIRKLPFQR LVREIAQDFK TDLRFQSSA VAALQEAAEA YLVGLFEDTN LCAIHAKRVT IMPKDIQLAR RIRGERA UniProtKB:  Histone H3.1 Histone H3.1 |
-Macromolecule #2: Histone H4
| Macromolecule | Name: Histone H4 / type: protein_or_peptide / ID: 2 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Arabidopsis thaliana (thale cress) Arabidopsis thaliana (thale cress) |
| Molecular weight | Theoretical: 11.436467 KDa |
| Recombinant expression | Organism:   Escherichia coli BL21 (bacteria) Escherichia coli BL21 (bacteria) |
| Sequence | String: MSGRGKGGKG LGKGGAKRHR KVLRDNIQGI TKPAIRRLAR RGGVKRISGL IYEETRGVLK IFLENVIRDA VTYTEHARRK TVTAMDVVY ALKRQGRTLY GFGG UniProtKB:  Histone H4 Histone H4 |
-Macromolecule #3: Histone H2A.6
| Macromolecule | Name: Histone H2A.6 / type: protein_or_peptide / ID: 3 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Arabidopsis thaliana (thale cress) Arabidopsis thaliana (thale cress) |
| Molecular weight | Theoretical: 13.680854 KDa |
| Recombinant expression | Organism:   Escherichia coli BL21 (bacteria) Escherichia coli BL21 (bacteria) |
| Sequence | String: MAGRGKTLGS GGAKKATSRS SKAGLQFPVG RIARFLKAGK YAERVGAGAP VYLAAVLEYL AAEVLELAGN AARDNKKTRI VPRHIQLAV RNDEELSKLL GDVTIANGGV MPNIHNLLLP KKAGASKPQE D UniProtKB: Histone H2A.6 |
-Macromolecule #4: Histone H2B.6
| Macromolecule | Name: Histone H2B.6 / type: protein_or_peptide / ID: 4 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Arabidopsis thaliana (thale cress) Arabidopsis thaliana (thale cress) |
| Molecular weight | Theoretical: 16.474459 KDa |
| Recombinant expression | Organism:   Escherichia coli BL21 (bacteria) Escherichia coli BL21 (bacteria) |
| Sequence | String: MAPRAEKKPA EKKPAAEKPV EEKSKAEKAP AEKKPKAGKK LPKEAGAGGD KKKKMKKKSV ETYKIYIFKV LKQVHPDIGI SSKAMGIMN SFINDIFEKL ASESSKLARY NKKPTITSRE IQTAVRLVLP GELAKHAVSE GTKAVTKFTS S UniProtKB: Histone H2B.6 |
-Macromolecule #7: ATP-dependent DNA helicase DDM1
| Macromolecule | Name: ATP-dependent DNA helicase DDM1 / type: protein_or_peptide / ID: 7 / Number of copies: 1 / Enantiomer: LEVO / EC number:  DNA helicase DNA helicase |
|---|---|
| Source (natural) | Organism:   Arabidopsis thaliana (thale cress) Arabidopsis thaliana (thale cress) |
| Molecular weight | Theoretical: 86.844836 KDa |
| Recombinant expression | Organism:   Escherichia coli BL21 (bacteria) Escherichia coli BL21 (bacteria) |
| Sequence | String: SMVSLRSRKV IPASEMVSDG KTEKDASGDS PTSVLNEEEN CEEKSVTVVE EEILLAKNGD SSLISEAMAQ EEEQLLKLRE DEEKANNAG SAVAPNLNET QFTKLDELLT QTQLYSEFLL EKMEDITING IESESQKAEP EKTGRGRKRK AASQYNNTKA K RAVAAMIS ...String: SMVSLRSRKV IPASEMVSDG KTEKDASGDS PTSVLNEEEN CEEKSVTVVE EEILLAKNGD SSLISEAMAQ EEEQLLKLRE DEEKANNAG SAVAPNLNET QFTKLDELLT QTQLYSEFLL EKMEDITING IESESQKAEP EKTGRGRKRK AASQYNNTKA K RAVAAMIS RSKEDGETIN SDLTEEETVI KLQNELCPLL TGGQLKSYQL KGVKWLISLW QNGLNGILAD QMGLGKTIQT IG FLSHLKG NGLDGPYLVI APLSTLSNWF NEIARFTPSI NAIIYHGDKN QRDELRRKHM PKTVGPKFPI VITSYEVAMN DAK RILRHY PWKYVVIDEG HRLKNHKCKL LRELKHLKMD NKLLLTGTPL QNNLSELWSL LNFILPDIFT SHDEFESWFD FSEK NKNEA TKEEEEKRRA QVVSKLHGIL RPFILRRMKC DVELSLPRKK EIIMYATMTD HQKKFQEHLV NNTLEAHLGE NAIRG QGWK GKLNNLVIQL RKNCNHPDLL QGQIDGSYLY PPVEEIVGQC GKFRLLERLL VRLFANNHKV LIFSQWTKLL DIMDYY FSE KGFEVCRIDG SVKLDERRRQ IKDFSDEKSS CSIFLLSTRA GGLGINLTAA DTCILYDSDW NPQMDLQAMD RCHRIGQ TK PVHVYRLSTA QSIETRVLKR AYSKLKLEHV VIGQGQFHQE RAKSSTPLEE EDILALLKED ETAEDKLIQT DISDADLD R LLDRSDLTIT APGETQAAEA FPVKGPGWEV VLPSSGGMLS SLNS UniProtKB: ATP-dependent DNA helicase DDM1 |
-Macromolecule #5: DNA (sense strand)
| Macromolecule | Name: DNA (sense strand) / type: dna / ID: 5 / Number of copies: 1 / Classification: DNA |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
| Molecular weight | Theoretical: 51.310664 KDa |
| Sequence | String: (DA)(DT)(DC)(DG)(DA)(DG)(DA)(DA)(DT)(DC) (DC)(DC)(DG)(DG)(DT)(DG)(DC)(DC)(DG)(DA) (DG)(DG)(DC)(DC)(DG)(DC)(DT)(DC)(DA) (DA)(DT)(DT)(DG)(DG)(DT)(DC)(DG)(DT)(DA) (DG) (DA)(DC)(DA)(DG)(DC)(DT) ...String: (DA)(DT)(DC)(DG)(DA)(DG)(DA)(DA)(DT)(DC) (DC)(DC)(DG)(DG)(DT)(DG)(DC)(DC)(DG)(DA) (DG)(DG)(DC)(DC)(DG)(DC)(DT)(DC)(DA) (DA)(DT)(DT)(DG)(DG)(DT)(DC)(DG)(DT)(DA) (DG) (DA)(DC)(DA)(DG)(DC)(DT)(DC)(DT) (DA)(DG)(DC)(DA)(DC)(DC)(DG)(DC)(DT)(DT) (DA)(DA) (DA)(DC)(DG)(DC)(DA)(DC)(DG) (DT)(DA)(DC)(DG)(DC)(DG)(DC)(DT)(DG)(DT) (DC)(DC)(DC) (DC)(DC)(DG)(DC)(DG)(DT) (DT)(DT)(DA)(DA)(DC)(DC)(DG)(DC)(DC)(DC) (DA)(DA)(DG)(DG) (DG)(DG)(DA)(DT)(DT) (DA)(DC)(DT)(DC)(DC)(DC)(DT)(DA)(DG)(DT) (DC)(DT)(DC)(DC)(DA) (DG)(DG)(DC)(DA) (DC)(DG)(DT)(DG)(DT)(DC)(DA)(DG)(DA)(DT) (DA)(DT)(DA)(DT)(DA)(DC) (DA)(DT)(DC) (DC)(DG)(DA)(DT)(DT)(DC)(DC)(DA)(DG)(DT) (DG)(DC)(DC)(DG)(DG)(DT)(DG) (DT)(DC) (DG)(DC)(DT)(DG)(DA) |
-Macromolecule #6: DNA (antisense strand)
| Macromolecule | Name: DNA (antisense strand) / type: dna / ID: 6 / Number of copies: 1 / Classification: DNA |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
| Molecular weight | Theoretical: 51.799977 KDa |
| Sequence | String: (DT)(DC)(DA)(DG)(DC)(DG)(DA)(DC)(DA)(DC) (DC)(DG)(DG)(DC)(DA)(DC)(DT)(DG)(DG)(DA) (DA)(DT)(DC)(DG)(DG)(DA)(DT)(DG)(DT) (DA)(DT)(DA)(DT)(DA)(DT)(DC)(DT)(DG)(DA) (DC) (DA)(DC)(DG)(DT)(DG)(DC) ...String: (DT)(DC)(DA)(DG)(DC)(DG)(DA)(DC)(DA)(DC) (DC)(DG)(DG)(DC)(DA)(DC)(DT)(DG)(DG)(DA) (DA)(DT)(DC)(DG)(DG)(DA)(DT)(DG)(DT) (DA)(DT)(DA)(DT)(DA)(DT)(DC)(DT)(DG)(DA) (DC) (DA)(DC)(DG)(DT)(DG)(DC)(DC)(DT) (DG)(DG)(DA)(DG)(DA)(DC)(DT)(DA)(DG)(DG) (DG)(DA) (DG)(DT)(DA)(DA)(DT)(DC)(DC) (DC)(DC)(DT)(DT)(DG)(DG)(DG)(DC)(DG)(DG) (DT)(DT)(DA) (DA)(DA)(DC)(DG)(DC)(DG) (DG)(DG)(DG)(DG)(DA)(DC)(DA)(DG)(DC)(DG) (DC)(DG)(DT)(DA) (DC)(DG)(DT)(DG)(DC) (DG)(DT)(DT)(DT)(DA)(DA)(DG)(DC)(DG)(DG) (DT)(DG)(DC)(DT)(DA) (DG)(DA)(DG)(DC) (DT)(DG)(DT)(DC)(DT)(DA)(DC)(DG)(DA)(DC) (DC)(DA)(DA)(DT)(DT)(DG) (DA)(DG)(DC) (DG)(DG)(DC)(DC)(DT)(DC)(DG)(DG)(DC)(DA) (DC)(DC)(DG)(DG)(DG)(DA)(DT) (DT)(DC) (DT)(DC)(DG)(DA)(DT) |
-Experimental details
-Structure determination
| Method |  cryo EM cryo EM |
|---|---|
 Processing Processing |  single particle reconstruction single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 2.2 mg/mL |
|---|---|
| Buffer | pH: 8 |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277.15 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD Bright-field microscopy / Cs: 2.7 mm / Nominal defocus max: 2.5 µm / Nominal defocus min: 1.0 µm Bright-field microscopy / Cs: 2.7 mm / Nominal defocus max: 2.5 µm / Nominal defocus min: 1.0 µm |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Detector mode: SUPER-RESOLUTION / Digitization - Frames/image: 1-32 / Number real images: 2858 / Average electron dose: 50.0 e/Å2 |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
-Atomic model buiding 1
| Refinement | Space: REAL |
|---|---|
| Output model |  PDB-8wh5: |
 Movie
Movie Controller
Controller





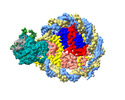

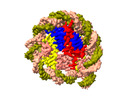


 Z
Z Y
Y X
X