+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-2429 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | The structure of the COPII coat assembled on membranes | |||||||||
 Map data Map data | sec31/13 outer COP2 coat layer vertex | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords |  COPII / coat / COPII / coat /  secretion / trafficking / secretion / trafficking /  Sec13 / Sec13 /  Sec31 Sec31 | |||||||||
| Function / homology |  : / : /  : :  Function and homology information Function and homology information | |||||||||
| Biological species |   Saccharomyces cerevisiae (brewer's yeast) Saccharomyces cerevisiae (brewer's yeast) | |||||||||
| Method | subtomogram averaging /  cryo EM / cryo EM /  negative staining / Resolution: 40.0 Å negative staining / Resolution: 40.0 Å | |||||||||
 Authors Authors | Zanetti G / Prinz S / Daum S / Meister A / Schekman R / Bacia K / Briggs JAG | |||||||||
 Citation Citation |  Journal: Elife / Year: 2013 Journal: Elife / Year: 2013Title: The structure of the COPII transport-vesicle coat assembled on membranes. Authors: Giulia Zanetti / Simone Prinz / Sebastian Daum / Annette Meister / Randy Schekman / Kirsten Bacia / John A G Briggs /  Abstract: Coat protein complex II (COPII) mediates formation of the membrane vesicles that export newly synthesised proteins from the endoplasmic reticulum. The inner COPII proteins bind to cargo and membrane, ...Coat protein complex II (COPII) mediates formation of the membrane vesicles that export newly synthesised proteins from the endoplasmic reticulum. The inner COPII proteins bind to cargo and membrane, linking them to the outer COPII components that form a cage around the vesicle. Regulated flexibility in coat architecture is essential for transport of a variety of differently sized cargoes, but structural data on the assembled coat has not been available. We have used cryo-electron tomography and subtomogram averaging to determine the structure of the complete, membrane-assembled COPII coat. We describe a novel arrangement of the outer coat and find that the inner coat can assemble into regular lattices. The data reveal how coat subunits interact with one another and with the membrane, suggesting how coordinated assembly of inner and outer coats can mediate and regulate packaging of vesicles ranging from small spheres to large tubular carriers. DOI:http://dx.doi.org/10.7554/eLife.00951.001. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_2429.map.gz emd_2429.map.gz | 7.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-2429-v30.xml emd-2429-v30.xml emd-2429.xml emd-2429.xml | 12.6 KB 12.6 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_2429.png emd_2429.png | 81.5 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-2429 http://ftp.pdbj.org/pub/emdb/structures/EMD-2429 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-2429 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-2429 | HTTPS FTP |
-Related structure data
| Related structure data |  2428C  2430C 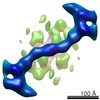 2431C  2432C  4bziC  4bzjC  4bzkC C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_2429.map.gz / Format: CCP4 / Size: 7.8 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_2429.map.gz / Format: CCP4 / Size: 7.8 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | sec31/13 outer COP2 coat layer vertex | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 4.3 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Sec13/31 complex (as part of complete COPII assembled on membrane...
| Entire | Name: Sec13/31 complex (as part of complete COPII assembled on membrane) cage vertex |
|---|---|
| Components |
|
-Supramolecule #1000: Sec13/31 complex (as part of complete COPII assembled on membrane...
| Supramolecule | Name: Sec13/31 complex (as part of complete COPII assembled on membrane) cage vertex type: sample / ID: 1000 / Oligomeric state: 4 heterotetramers form a vertex / Number unique components: 2 |
|---|---|
| Molecular weight | Experimental: 319.236 KDa / Theoretical: 319.236 KDa |
-Macromolecule #1: Sec31
| Macromolecule | Name: Sec31 / type: protein_or_peptide / ID: 1 / Number of copies: 4 / Oligomeric state: heterotetramer / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:   Saccharomyces cerevisiae (brewer's yeast) / synonym: Baker's yeast / Location in cell: cytosol/endoplasmic reticulum Saccharomyces cerevisiae (brewer's yeast) / synonym: Baker's yeast / Location in cell: cytosol/endoplasmic reticulum |
| Molecular weight | Experimental: 138.824 KDa / Theoretical: 138.824 KDa |
| Recombinant expression | Organism:   Saccharomyces cerevisiae (brewer's yeast) / Recombinant strain: RSY1112 / Recombinant plasmid: pNS3141 (6H31/CK1313) Saccharomyces cerevisiae (brewer's yeast) / Recombinant strain: RSY1112 / Recombinant plasmid: pNS3141 (6H31/CK1313) |
| Sequence | UniProtKB:  UNIPROTKB: E7Q1I6 UNIPROTKB: E7Q1I6 |
-Macromolecule #2: Sec13
| Macromolecule | Name: Sec13 / type: protein_or_peptide / ID: 2 / Number of copies: 4 / Oligomeric state: heterotetramer / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:   Saccharomyces cerevisiae (brewer's yeast) / synonym: Baker's yeast / Location in cell: cytosol/endoplasmic reticulum Saccharomyces cerevisiae (brewer's yeast) / synonym: Baker's yeast / Location in cell: cytosol/endoplasmic reticulum |
| Molecular weight | Experimental: 20.79 KDa / Theoretical: 27.9 KDa |
| Recombinant expression | Organism:   Saccharomyces cerevisiae (brewer's yeast) / Recombinant strain: RSY1112 / Recombinant plasmid: pNS3141 (6H31/CK1313) Saccharomyces cerevisiae (brewer's yeast) / Recombinant strain: RSY1112 / Recombinant plasmid: pNS3141 (6H31/CK1313) |
| Sequence | UniProtKB:  UNIPROTKB: E7Q6Z3 UNIPROTKB: E7Q6Z3 |
-Experimental details
-Structure determination
| Method |  negative staining, negative staining,  cryo EM cryo EM |
|---|---|
 Processing Processing | subtomogram averaging |
| Aggregation state | helical array |
- Sample preparation
Sample preparation
| Concentration | 0.03 mg/mL |
|---|---|
| Buffer | pH: 6.8 / Details: HEPES, 50 mM KOAc, 1.2 mM MgCl2 |
| Staining | Type: NEGATIVE / Details: plunge frozen |
| Grid | Details: C-flat grids |
| Vitrification | Cryogen name: ETHANE / Instrument: HOMEMADE PLUNGER |
- Electron microscopy #1
Electron microscopy #1
| Microscope | FEI TITAN KRIOS |
|---|---|
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD Bright-field microscopy / Cs: 2.7 mm / Nominal defocus max: 3.2 µm / Nominal defocus min: 2.0 µm / Nominal magnification: 19500 Bright-field microscopy / Cs: 2.7 mm / Nominal defocus max: 3.2 µm / Nominal defocus min: 2.0 µm / Nominal magnification: 19500 |
| Specialist optics | Energy filter - Name: GATAN GIF 2002 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Tilt series - Axis1 - Min angle: -60 ° / Tilt series - Axis1 - Max angle: 60 ° |
| Microscopy ID | 1 |
| Date | Sep 18, 2012 |
| Image recording | Category: CCD / Film or detector model: GATAN MULTISCAN / Number real images: 26 / Average electron dose: 80 e/Å2 / Bits/pixel: 16 |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Electron microscopy #2
Electron microscopy #2
| Microscope | FEI TITAN KRIOS |
|---|---|
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD Bright-field microscopy / Cs: 2.7 mm / Nominal defocus max: 3.2 µm / Nominal defocus min: 2.0 µm / Nominal magnification: 19500 Bright-field microscopy / Cs: 2.7 mm / Nominal defocus max: 3.2 µm / Nominal defocus min: 2.0 µm / Nominal magnification: 19500 |
| Specialist optics | Energy filter - Name: GATAN GIF 2002 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Tilt series - Axis1 - Min angle: -60 ° / Tilt series - Axis1 - Max angle: 60 ° |
| Microscopy ID | 2 |
| Date | Jun 19, 2012 |
| Image recording | Category: CCD / Film or detector model: GATAN MULTISCAN / Number real images: 26 / Average electron dose: 80 e/Å2 / Bits/pixel: 16 |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| CTF correction | Details: each tilted image within tomogram |
|---|---|
| Final angle assignment | Details: 0 0 0 in zyz convention |
| Final reconstruction | Applied symmetry - Point group: C2 (2 fold cyclic ) / Algorithm: OTHER / Resolution.type: BY AUTHOR / Resolution: 40.0 Å / Resolution method: FSC 0.5 CUT-OFF / Software - Name: TOM/AV3, Matlab, dynamo / Number subtomograms used: 417 ) / Algorithm: OTHER / Resolution.type: BY AUTHOR / Resolution: 40.0 Å / Resolution method: FSC 0.5 CUT-OFF / Software - Name: TOM/AV3, Matlab, dynamo / Number subtomograms used: 417 |
| Details | see materials and methods in relevant publication |
 Movie
Movie Controller
Controller









